Jin Kim Montclare
,
Ph.D.
-
Professor of Chemical and Biomolecular Engineering

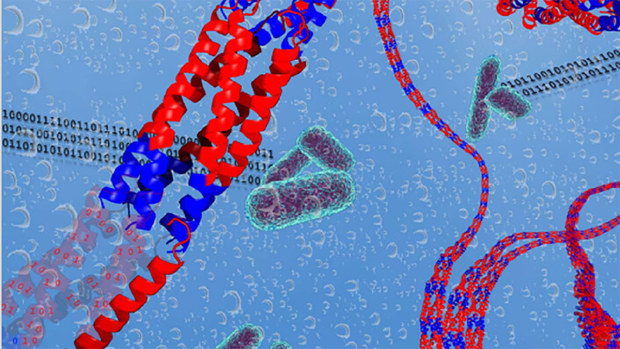
Jin Kim Montclare is a Professor in the Department of Chemical and Biomolecular Engineering, who is performing groundbreaking research in engineering proteins to mimic nature and, in some cases, work better than nature. She works to customize artificial proteins with the aim of targeting human disorders, drug delivery and tissue regeneration as well as create nanomaterials for electronics. Using multidisciplinary expertise in chemistry and genetic engineering, these results have already been realized.
Prior to joining NYU-Tandon, Montclare was a postdoctoral fellow at the California Institute of Technology in the Division of Chemistry and Chemical Engineering.
She received a Bachelor of Science in Chemistry from Fordham University in 1997, a Master of Science and a Ph.D. in Bioorganic Chemistry from Yale University in 2001 and 2003, respectively.
Among her many honors and awards are the AAAS Leshner Fellowship, AIMBE Fellow, ACS Rising Star Award, Agnes Faye Morgan Research Award from Iota Sigma Pi, Executive Leadership in Academic Technology and Engineering Fellowship, American Chemical Society PROGRESS /Dreyfus Lectureship, the Dreyfus Special Grants Program Award, the Air Force Office of Scientific Research Young Investigator Award, the Wechsler Award for Excellence, the Othmer Junior Fellow Award, the National Institute’s of Health Postdoctoral Fellowship, and the National Science Foundation Pre-doctoral Fellowship.
Montclare is the author of numerous papers for refereed journals, colloquia, and seminars and holds several patents.
She is a member of the American Chemical Society, the International Society for Pharmaceutical Engineering, the Biophysical Society, the Materials Research Society, the Biochemical Society, the Protein Society and American Association of Cancer Research, and the American Institute of Chemical Engineers.
Associated websites and social media
Education
Yale University, 2003
MS, Ph.D., Bioorganic Chemistry
Fordham University, 1997
BS, Chemistry
Experience
NYU Tandon School of Engineering
Professor
The Protein Engineering and Molecular Design Lab began July 2005. Broadly, our lab is focused on engineering macromolecules. The long-term goal of our lab research is to be able to predictably design or engineer artificial therapeutics, biocatalysts, scaffolds and cells. We seek to provide biologically inspired solutions to address the challenges of human disorder treatment and medicine, sustainable energy and environmental remediation.
From: July 2005 to present
California Institute of Technology
Postdoctoral fellow
Worked in the laboratory of Professor David Tirrell on evolving proteins bearing unnatural amino acids.
From: January 2003 to July 2005
Publications
Journal Articles
-
- Lindsay K. Hill,† Michael Meleties,† Xuan Xie, Erika Delgado-Fukushima, Teeba Jihad, Che Fu Liu, Sean O’Neill, Raymond S. Tu, P. Douglas Renfrew, Richard Bonneau, Youssef Z. Wadghiri & Jin K. Montclare. Thermoresponsive Protein-Engineered Coiled-coil Hydrogel for Sustained Small Molecule Release. Biomacromolecules (2019) 20, 3340-3351. DOI: https://doi.org/10.1021/acs.biomac.9b00107
- Priya Katyal, Michael Meleties & Jin K. Montclare. Self-Assembled Protein and Peptide Nanomaterials. ACS Biomaterials Science & Engineering (2019) https://doi.org/10.1021/acsbiomaterials.9b00408
- Kamia Punia, Jacob Kronenberg & Jin K. Montclare. Protein Materials for Theranostic Applications. Molecular Systems Design & Engineering (2019) 4, 1074 – 1094. doi: 10.1039/C9ME00143C
- Lindsay K. Hill, Joseph A. Frezzo, Priya Katyal, Dung Minh Hoang, Zakia Ben Youss Gironda, Cynthia Xu, Xuan Xie, Erika Delgado-Fukushima, Youssef Z. Wadghiri & Jin K. Montclare. Protein Engineered Nanoscale Micelles for Dynamic 19F Magnetic Resonance and Therapeutic Drug Delivery. ACS Nano (2019) doi:10.1021/acsnano.8b07481
- Yao Wang, Priya Katyal & Jin K. Montclare. Protein Engineered Functional Materials. Advanced Healthcare Materials (2019) doi: 10.1002/adhm.201801374.
- Hironori Kasai, Kenji Inoue, Kentaro Imamura, Carlo Yuvienco, Jin K Montclare & Seiichi Yamano. Efficient siRNA delivery and gene silencing using a lipopolypeptide hybrid vector mediated by a caveolae-mediated and temperature-dependent endocytic pathway. Journal of Nanobiotechnology (2019) 17:11 https://doi.org/10.1186/s12951-019-0444-8
- Liming Yin, Albert S. Agustinus, Carlo Yuvienco, Takeshi Minashima, Nicole L. Schnabel, Thorsten Kirsch & Jin K. Montclare. Engineered Coiled-Coil Protein for Delivery of Inverse Agonist for Osteoarthritis. Biomacromolecules (2018) 19, 1614-1624. DOI: 10.1021/acs.biomac.8b00158
- Andrew J. Olsen, Jennifer S. Haghpanah, Priya Katyal, Matthew B. Kubilius, Ruipeng Li, Nicole L. Schnabel, Sean C. O’Neill, Yao Wang, Min Dai, Navjot Singh, Raymond S. Tu & Jin K. Montclare. Protein Engineered Triblock Polymers Comprised of Two SADs: Enhanced Mechanical Properties and Binding Abilities. Biomacromolecules (2018) 19, 1552-1561. DOI: 10.1021/acs.biomac.7b01259
- Andrew J. Olsen, Leif A. Halvorsen, Ching-Yao Yang, Roni Barak Ventura, Liming Yin, P. Douglas Renfrew,, Richard Bonneau & Jin K. Montclare. Impact of Phenylalanines Outside the Dimer Interface on Phophotriesterase Stability and Function. Mol BioSys (2017) 13, 2092-2106. DOI: 10.1039/c7mb00196g
- Che Fu Liu, Raymond Chen, Joseph A. Frezzo, Priya Katyal, Lindsay K. Hill, Liming Yin, Nikita Srivastava, Haresh T. More, P. Douglas Renfrew, Richard Bonneau & Jin K. Montclare. Efficient dual siRNA and drug delivery using engineered lipoproteoplexes. Biomacromolecules (2017) 18, 2688-2698. DOI: 10.1021/acs.biomac.7b00203.
- Liming Yin, Carlo Yuvienco & Jin K. Montclare. Protein based therapeutic delivery agents: Contemporary developments and challenges. Biomaterials (2017) 134, 91-116. DOI: https://doi.org/10.1016/j.biomaterials.2017.04.036
- Piul S Rabbani, Anna Zhou, Zachary M Borab, Joseph A Frezzo, Nikita Srivastava, Haresh T More, William J Rifkin, Joshua A David, Samuel J Berens, Raymond Chen, Sophia Hameedi, Muhammad H Junejo, Camille Kim, Rita A Sartor, Che F Liu, Pierre B Saadeh, Jin K Montclare & Daniel J Ceradini. Novel lipoproteoplex delivers Keap1 siRNA based gene therapy to accelerate diabetic wound healing. Biomaterials (2017) 132, 1-15. DOI: http://dx.doi.org/10.1016/j.biomaterials.2017.04.001
- Min Dai,† Joseph A. Frezzo,† Etka Sharma, Raymond Chen, Navjot Singh, Carlo Yuvienco, Elif Caglar, Shu Xiao, Anjana Saxena & Jin K. Montclare. Engineered Protein Polymer-Gold Nanoparticle Hybrid Materials for Small Molecule Delivery. J Nanomed Nanotechnol (2016) 7:356. doi:10.4172/2157-7439.1000356
- Jasmin Hume, Raymond Chen, Rudy Jacquet, Michael Yang & Jin K. Montclare. Tunable Conformation Dependent Protein•Gold Nanoparticle Nanocomposites. Biomacromolecules (2015) 16, 1706-1713. DOI: 10.1021/acs.biomac.5b00098
- Joseph A. Frezzo & Jin K. Montclare. Exploring the potential of engineered coiled-coil protein microfibers in drug delivery. Therapeutic Delivery (2015) 6, 643-646.
- Haresh T. More, Kevin S. Zhang, Nikita Srivastiva, Joseph A. Frezzo & Jin K. Montclare. Influence of Fluorination on Protein Engineered Coiled-coil Fibers. Biomacromolecules (2015) 16, 1210–1217. DOI: 10.1021/bm5019062
- Jasmin Hume, Jennifer Sun, Rudy Jacquet, P. Douglas Renfrew, Jesse A. Martin, Richard Bonneau, M. Lane Gilchrist & Jin K. Montclare. Engineered Coiled-Coil Protein Microfibers. Biomacromolecules (2014) 15, 3503-3510. DOI: 10.1021/bm5004948
- Ching-Yao Yang, P. Douglas Renfrew, Andrew J. Olsen, Michelle Zhang, Carlo Yuvienco, Richard Bonneau, Jin K. Montclare. Improved Stability and Half-life of Fluorinated Phosphotriesterase using Rosetta. ChemBioChem (2014) 15, 1761-1764.
- Haresh T. More, Joseph A. Frezzo, Jisen Dai, Seiichi Yamano, Jin K. Montclare. Efficient Gene Delivery from Supercharged Coiled-coil Protein and Cationic Lipid Nanocomplexes. Biomaterials (2014) 35, 7188-7193.
- Seiichi Yamano, Jisen Dai, Shigeru Hanatani, Ken Haku, Takuto Yamanaka, Mika Ishioka, Tadahiro Takayama, Carlo Yuvienco, Sachin Khapli, Amr M. Moursi, Jin K. Montclare. Long-term efficient gene delivery using polyethylenimine with modified Tat peptide. Biomaterials (2014) 34, 1705-1715.
- Jennifer S. Haghpanah, Raymond Tu, Sandra Da Silva, Deng Yan, Silvana Mueller, Christoph Weder, E. Johan Foster, Iulia Sacui, Jeffery W. Gilman, Jin K. Montclare. Bionanocomposites: Differential Effects of Cellulose Nanocrystals on Protein Diblock Copoloymers. Biomacromolecules (2013) 14, 4360-4367.
- Daniel Yoo, Nick Tovar, Ryo Jimbo, Charles Marin, Rodolfo Anchieta, Lucas Machado, Jin Montclare, Fernando Guastaldi, Malvin Janal & Paulo Coelho, Increased Osseointegration Effect of BMP-2 on Dental Implants: An In Vivo Study. Clinical Implant Dentistry and Related Research. (2013) doi: 10.1002/jbm.a.34862
- Herbert Lannon, Jennifer S. Haghpanah, Jin K. Montclare, Eric Vanden-Eijnden & Jasna Brujic. Force-clamp experiments reveal the free energy profile and diffusion coefficient of the collapse of proteins. Phys, Rev. Lett. (2013) 110, 128301-6. doi:10.1103/PhysRevLett.110.128301
- Nancy Hom, Kinjal R. Mehta, Tsengming Chou, Amy B. Foraker, Frances M. Brodsky, Kent Kirshenbaum & Jin K. Montclare. Anisotropic nanocrystal arrays organized on protein lattices formed by recombinant clathrin fragments. J. Mat. Chem. (2012) 22, 23335-23339.
- Carlo Yuvienco, Haresh T. More, Jennifer S. Haghpanah, Raymond S. Tu & Jin K. Montclare, Modulating Supramolecular Assemblies and Mechanical Properties of Engineered Protein Materials by Fluorinated Amino Acids. Biomacromolecules. (2012) 13, 2273-2278.
- Susheel K. Gunasekar, Luona Anjia, Hiroshi Matsui & Jin K. Montclare. Effects of Divalent Metals on Nanoscopic Fiber Formation and Small Molecule Recognition of Helical Proteins. Adv. Funct. Mat. (2012) 22, 2154-2159.
- Peter J. Baker, Yan M. Chan, Moritz Hertel & Jin K. Montclare. Characterization and Identification of Protein Partners of Fn3 Domain in FnTm2. Protein Expression and Purification (2012) 81, 42-48.
- Peter J. Baker, Christopher S. Poultney, Zhiqiang Liu, Richard Gross & Jin K. Montclare. Identification and Comparison of Cutinases for Synthetic Polyester Degradation. Applied Microbiology and Biotechnology (2012) 93, 229-240.
- Min Dai, jennifer S. Haghpanah, Navjot Singh, Eric W. Roth, Alice Liang, Raymond S. Tu & Jin K. Montclare, Artificial Protein Block Polymer Libraries Bearing Two SADs: Effects of Elastin Domain Repeats. Biomacromolecules. (2011) 12, 4240-4246.
- Kinjal R. Mehta, Ching-Yao Yang & Jin K. Montclare, Modulating substrate specificity of histone acetyltransferase with unnatural amino acids. Mol. BioSyst. (2011) 7, 3050-3055
- Peter J. Baker & Jin K. Montclare, Enhanced refoldability and thermoactivity of fluorinated phosphotriesterase. Chembiochem, (2011) 12, 1845-1848.
- Seiichi Yamano, Jisen Dai, Carlo Yuvienco, Sachin Khapli, Amr M. Moursi & Jin K. Montclare. Modified Tat peptide with lipids enhances gene transfection efficiency via temperature-dependent and caveolae-mediated endocytosis. J. Contr. Rel. (2011) 152, 278-285.
- Jennifer S. Haghpanah, Carlo Yuvienco, Eric W. Roth, Alice Liang, Raymond S. Tu & Jin K. Montclare, Supramolecular Assembly and Small Molecule Recognition by Genetically Engineered Protein Block Polymers Composed of Two SADs. Mol. BioSys. (2010) 6, 1662-1667.
- Kinjal R. Mehta, Yan M. Chan+, Man X. Lee+, Ching Yao Yang, Natalya Voloshchuk & Jin K. Montclare. Mutagenesis of tGCN5 core region reveals two critical surface residues F90 and R140. Biochem. Biophys. Res. Comm. (2010) 400, 363-368.
- Natalya Voloshchuk & Jin K. Montclare, Incorporation of unnatural amino acids for synthetic biology. Mol. BioSys. (2010) 6, 65-80.
- Jennifer S. Haghpanah, Carlo Yuvienco, Deniz E. Civay, Hanna Barra, Peter J. Baker, Sachin Khapli, Natalya Voloshchuk, Susheel K. Gunasekar, Murugappan Muthukumar & Jin K. Montclare, Artificial protein block copolymers comprised of two self-assembling domains. ChemBioChem. (2009) 10, 2733-2735. *Highlighted in Futurity* http://futurity.org/
- Zhiqiang Liu, Yuying Gosser, Peter James Baker, Yaniv Ravee, Ziying Lu, Girum Alemu, Huiguang Li, Glenn L. Butterfoss, Xiang-Peng Kong, Richard Gross & Jin K. Montclare, Structural and functional studies of A. oryzae cutinase: Enhanced thermostability and hydrolytic activity of synthetic ester and polyester degradation. J. Am. Chem Soc. (2009) 131, 15711-15716.
- Susheel K. Gunasekar, Mukta Asnani, Chandani Limbad, Jennifer S. Haghpanah, Wendy Hom, Hanna Barra, Soumya Nanda & Jin K. Montclare, N-terminal aliphatic residues dictate the structure, stability and assembly of the coiled-coil region of COMP. Biochemistry. (2009) 48, 8559–8567.
- Natalya Voloshchuk, Anita Yuhua Zhu, David Snydacker & Jin K. Montclare, Positional effects of monofluorinated phenylalanines on histone acetyltransferase stability and activity. Bioorg. Med. Chem. Lett. (2009) 19, 5449-5451.
- Sachin Khapli, Jin R. Kim, Jin K. Montclare, Rastislav Levicky, Mauricio Porfiri and Stavroula Sofou. Frozen cyclohexane-in-water emulsion as a sacrificial template for the synthesis of multilayered polyelectrolyte microcapsules. Langmuir, (2009) 17, 9728-9733.
- Jin K. Montclare*, Soojin Son*, Ginevra Clark, Krishna Kumar & David A. Tirrell, Biosynthesis of stable dimeric coiled-coils bearing (2S, 4R)-5’,5’,5’-trifluoroleucine and (2S, 4S)-5’,5’,5’-trifluoroleucine. ChemBioChem (2009) 10, 84-86. * these authors contributed equally
- Susheel K. Gunasekar, Jennifer S. Haghpanah & Jin K. Montclare, Assembly of bioinspired protein fibers. Polymers for Advanced Technology. (2008) 19, 454-468.
- Natalya Voloshchuk, Man Xia Lee, Wan Wen Zhu, Ismet Caglar Tanrikulu & Jin K. Montclare, Fluorinated chloramphenicol acetyltransferase thermostability and activity profile: improved thermostability by a single-isoleucine mutant. Bioorganic Medicinal Chemistry Letters (2007) 17, 5907-5911.
- Tatyana Panchenko, Wan Wen Zhu & Jin K. Montclare, Influence of global fluorination on chloramphenicol acetyltransferase activity and stability. Biotechnology and Bioengineering (2006) 94, 921-930.
- Jin K. Montclare, Soojin Son, Ginevra Clark, Krishna Kumar & David A. Tirrell, Biosynthesis of stable dimeric coiled-coils bearing (2S, 4R)-5’,5’,5’-trifluoroleucine and (2S, 4S)-5’,5’,5’-trifluoroleucine. ChemBioChem (2009) 10, 84-86.
- Jin K. Montclare & David A. Tirrell, Evolving proteins of novel composition. Angewandte Chemie International Edition (2006) 45, 4518-4521.
- Jin K. Montclare & Alanna Schepartz, Miniature homeodomains: High specificity without an N-terminal arm. J. Am. Chem. Soc. (2003) 125, 3416-3417.
- Jin K. Montclare, Leslie S. Sloan & A. Schepartz, Electrostatic control of half-site spacing preferences by the cyclic AMP Response Element Binding Protein: Selectivity at the expense of affinity. Nucleic Acids Res. (2001) 29, 3311-3319.
Authored/Edited Books
Joseph A. Frezzo & Jin K. Montclare, Natural Composite Systems for Bioinspired Materials. In Protein-based Engineered Nanostructures (2015) Edited by T. Z. Groves and A. L. Cortajarena. In press.
Haresh T. More, Ching-Yao Yang & Jin K. Montclare, Post-Translational Modification of Proteins Incorporating Non-natural Amino Acids. In Functional Polymers by Post-Polymerization Modification: Concepts, Practical Guidelines and Applications (2012) Edited by H. A. Klok and P. Theato.
Peter James Baker, Jennifer S. Haghpanah & Jin K. Montclare Elastin-basted Protein Polymers. In Polymer Biocatalysts and Biomaterials II (2008) Edited by H. N. Cheng and R. A. Gross. Chapter 3.
Peter J. Baker & Jin K. Montclare, Biotransformations using Cutinase. In Green Polymer Chemistry: Biocatalysis and Biomaterials (2010) Edited by H. N. Cheng and R. A. Gross. 1043, 141-158.
Other Publications
Priya Chacko, Sarah Appelbaum, Heejoo Kim, Jinhui Zhao & Jin K. Montclare. Integrating Technology in STEM Education. J. Tech. Sci. Ed. (2015) 5, 5-14.
Heejoo Kim, Priya Chacko, Jinhui Zhao & Jin K. Montclare. Using Touch-Screen Technology, Apps, and Blogs To Engage and Sustain High School Students’ Interest in Chemistry Topics. J. Chem. Ed. (2014) 91, 1818-1822. doi: 10.1021/ed500234z.
Maurica S. Lewis, Jinhui Zhao & Jin K. Montclare. Development and Implementation of High School Chemistry Modules using Touch-Screen Technologies. J. Chem. Ed. (2012) 89, 1012-1018.
Robert Lorenzini, Maurica S. Lewis & Jin K. Montclare. College-Mentored Polymer/Materials Science Modules for Middle and High School Students. J. Chem. Ed. (2011) 88, 1105-1108.
Yan M. Chan, Wendy Hom & Jin K. Montclare. Mentored Chem-Bio Technology Lab to Promote Early Interest in Science. J. Chem. Ed. (2011) 88, 751-754.
Affiliations
General/Collaborative Research
- CBAS group: Jin Kim, Rasti Levicky, Maurizio Porfiri, NYU-Poly
- Thorsten Kirsch, NYU Med
- Daniel Ceradini, NYU Med
- Vikram Kapila, NYU-Poly
- Sungheon Kim, NYU Med
- Komal Jhaveri, NYU Med
- Youssef Wadghiri, NYU Med
- Kent Kirshenbaum, Chemistry, NYU
- Jasna Brujic, Physics, NYU
- Xiang-Peng Kong, NYU Med
- Seiichi Yamano, NYU Dental
- Paulo Coelho, NYU Dental
Affiliations
SUNY Downstate Medical Center Rockefeller University
NYU Chemistry
Awards
- 2016 ACS WCC Rising Star Award
- 2015 Agnes Fay Morgan Research Award from Iota Sigma Pi, National Honor Society for Women in Chemistry
- 2014 Distinguished Award for Excellence, Dedication to Invention, Innovation and Entrepreneurship
- 2014 Executive Leadership in Academic Technology and Engineering Fellow
- 2013 NSF I-Corps
- 2009 Young Observer Fellowship (declined)
- 2008 ACS PROGRESS/Dreyfus Lectureship
- 2008 Dreyfus Special Grants Program Award
- 2007 AFOSR Young Investigator Award
- 2006 Wechsler Award for Excellence
- 2006 Othmer Junior Fellow, Othmer Institute
- 2003-2005 National Institute of Health Postdoctoral Fellowship
- 2001 T. F. Cooke Teaching Award for Organic Chemistry; Yale University
- 1997-2000 National Science Foundation Graduate Research Fellowship
- 1997-2000 Pfizer Fellowship
Grants
Engineered Protein-Lipid Systems for siRNA and Small Molecule Delivery, (PI)
National Science Foundation, 09/01/15-08/31/18
Patterned protein and hybrid materials: responsive ‘chemomechanical’ shape-shifters, (PI)
Army Research Offcie, 08/01/15-07/31/18
PFI-AIR: Prototyping a Gene Transfection Tool, GeneTrain, (PI)
National Science Foundation, 08/01/14-12/31/15
Bottom-up Assembly of Engineered Protein Fibers, (PI)
Army Research Office, 10/01/11-09/30/14
Engineered Protein-Based Multi-Functional Materials, (PI)
National Science Foundation, 09/01/12-08/31/15
Computational Studies of Histone Modifications, (co-I)
National Institute of Health, 07/01/13-06/30/17
Engineered Protein Polymers, (PI)
AFOSR, 2007-10
Information for Mentees
Mentoring Style: informal and can listen and provide whatever feedback is needed; member of T&P Committee
My Research: Protein Engineering, Biomaterials, Diversity in Entrepreneurship
Research News
Self-assembling proteins can be used for higher performance, more sustainable skincare products
If you have a meticulous skincare routine, you know that personal skincare products (PSCPs) are a big business. The PSCP industry will reach $74.12 billion USD by 2027, with an annual growth rate of 8.64%. With such competition, companies are always looking to engineer themselves an edge, producing products that perform better without the downsides of current offerings.
In a new study published in ACS Applied Polymer Materials from the lab of Professor of Chemical and Biomolecular Engineering Jin Kim Montclare, researchers have created a novel protein-based gel as a potential ingredient in sustainable and high-performance PSCPs. This protein-based material, named Q5, could transform the rheological — or flow-related — properties of PSCPs, making them more stable under the slightly acidic conditions of human skin. This innovation could also streamline the creation of more eco-friendly skincare products, offering increased efficacy and durability while addressing market demands for ethically sourced ingredients.
Personal skincare products, ranging from beauty cosmetics to medical creams, rely on sophisticated “chassis” formulations — often emulsions or gels — to effectively deliver active ingredients. The performance of these products depends heavily on the stability and responsiveness of their chassis under various environmental conditions, particularly pH.
Current formulations often rely on ingredients such as polysaccharides or synthetic polymers to achieve the desired texture, stability, and compatibility with skin's natural pH, which is mildly acidic (most human skin has a pH of between 5.4–5.9). However, these traditional rheological modifiers have raised environmental concerns regarding sourcing and sustainability.
To take on this challenge, Montclare and her colleagues fabricated a self-assembling coiled-coil protein they call Q5. In the study, Q5 demonstrated impressive pH stability. The protein's unique structure enables it to form strong gels that do not degrade easily under acidic conditions, enhancing the longevity and performance of skincare products.This resilience marks a significant improvement over earlier protein-based gels, which typically disassemble in lower pH environments.
Notably, the research suggests that Q5 could be produced sustainably via bacterial or yeast fermentation, circumventing the ethical and ecological issues associated with animal-derived proteins or synthetic polymers. The protein’s natural amphiphilicity — its ability to attract and retain moisture — also enables it to bind various molecules, adding versatility as a moisturizer or binding agent in skincare products.
The research suggests that these protein-based rheological modifiers like Q5 could soon become a valuable component in the next generation of high-performance skincare products, helping brands meet consumer demand for sustainable beauty solutions without compromising on quality or functionality.
Britton, D., Sun, J., Faizi, H. A., Yin, L., Gao, W., & Montclare, J. K. (2024). Recombinant fibrous protein gels as rheological modifiers in skin ointments. ACS Applied Polymer Materials, 6(20), 12832–12841. https://doi.org/10.1021/acsapm.4c02468
Researchers harnessing exosomes and hydrogels for advanced diabetic wound healing
Diabetes, a widespread condition affecting approximately 13% of American adults, is often accompanied by complications such as impaired wound healing. If left unchecked, this can lead to severe outcomes, including the need for amputation. The challenge of finding effective treatments for diabetic wounds has grown increasingly urgent. Such wounds are marked by prolonged inflammation, lack of oxygen, and disrupted blood vessel formation, which all contribute to delayed recovery. However, a new frontier in biomedical research is pointing toward exosomes as a potential solution.
A team from NYU Langone and NYU Tandon including Jin Kim Montclare has began to explore exosomes, tiny membrane-bound vesicles, as promising tools for healing. These nanovesicles carry various biological materials — nucleic acids, proteins, and lipids — allowing them to mediate intercellular communication and influence processes such as tissue repair.
Specifically, exosomes derived from mesenchymal stem cells (MSCs), including those from adipose tissue, have demonstrated significant potential in promoting wound healing in animal models. Their therapeutic effects appear to stem from their ability to reduce inflammation and promote a healing-friendly environment by enhancing blood vessel formation and encouraging the activity of cells like fibroblasts and endothelial cells, which are essential for tissue repair.
One major advantage of exosomes is their ability to bypass some of the risks associated with traditional stem cell therapies, such as uncontrolled cell growth or immune rejection. However, despite their promise, exosomes typically require repeated administration—either through subcutaneous or intravenous injections—which poses a challenge for long-term wound management.
Montclare’s team have been exploring innovative ways to enhance the therapeutic potential of exosomes, one of which involves combining them with hydrogels. Hydrogels, composed of networks of cross-linked polymers, can encapsulate exosomes within their structure. This encapsulation enables a more sustained and localized release of exosomes directly at the wound site, without the need for invasive injections.
Hydrogels are already recognized for their biocompatibility and ability to hydrate wounds, making them useful as wound dressings on their own. When combined with exosomes, their therapeutic effectiveness increases significantly, especially for diabetic wounds.
Recent studies have shown that hydrogel-exosome combinations consistently lead to faster wound closure than either hydrogels or exosomes used alone. These hydrogel systems are not protein-based, but recent advances in protein-based hydrogel technology have opened new possibilities for improving wound healing.
Montclare has developed a protein-based hydrogel, referred to as "Q," which forms a gel at low temperatures through a process called upper critical solution temperature (UCST) gelation. This protein-based hydrogel self-assembles into nanofibers, forming a physically cross-linked network that provides mechanical strength. By fine-tuning the protein sequence using advanced computational tools, such as the Rosetta score and Poisson-Boltzmann electrostatic potential calculations, they have been able to improve the gel’s mechanical properties, stability, and speed of formation — key factors in creating an ideal wound dressing.
To push this approach further, they designed a variant of the Q hydrogel, dubbed Q5, using automated selection methods to optimize its stability. They encapsulated exosomes within Q5 to create a new hydrogel-exosome system, called Q5Exo. This system offers a topical, noninvasive wound dressing that holds promise for treating diabetic wounds more effectively than traditional methods, which rely on injections.
In studies using diabetic mouse models, Q5Exo demonstrated a significant reduction in healing time when applied topically compared to exosomes administered via injection. This suggests that protein-based hydrogels, with their tunable properties, could become a powerful platform for enhancing wound healing outcomes in diabetes. As research continues, such hydrogels could pave the way for a new generation of biocompatible, efficient wound dressings that harness the therapeutic power of exosomes.
Exosome Loaded Protein Hydrogel for Enhanced Gelation Kinetics and Wound Healing; Dustin Britton, Dianny Almanzar, Yingxin Xiao, Hao-Wei Shih, Jakub Legocki, Piul Rabbani, and Jin Kim Montclare; ACS Applied Bio Materials; 2024 7 (9), 5992-6000
Cutting-edge cancer treatments research promises more effective interventions
Cancer is one of the most devastating diagnoses that a person can receive, and it is a society-wide problem. According to the National Cancer Institute, an estimated 2,001,140 new cases of cancer will be diagnosed in the United States and 611,720 people will die from the disease this year. While cancer treatments have seen large improvements over the last decades, researchers are still laser-focused on developing strategies to defeat cancers, especially those that have been resistant to traditional interventions.
New research from Jin Kim Montclare, Professor of Chemical and Biomolecular Engineering, may lend hope to cancer patients in the future. Montclare’s lab, which uses customized artificial proteins to target human disorders, drug delivery and tissue regeneration utilizing a blend of chemistry and genetic engineering, has recently published two papers that take aim at cancers that have been difficult to treat.
MAPping a course to recovery
In the ongoing quest to develop more effective cancer therapies, these researchers have unveiled a promising approach utilizing protein-based targeting agents. A recent study published in Biomaterials Science introduces a novel strategy that harnesses the power of multivalent assembled proteins (MAPs) to target hypoxic tumors with unprecedented precision and efficacy.
Traditional cancer treatments often rely on passive or active targeting mechanisms to deliver therapeutic agents to tumor sites. However, these approaches have limitations, particularly in overcoming physiological and pathological barriers. To address these challenges, the research team focused on exploiting the unique features of the tumor microenvironment (TME), specifically its hypoxic conditions.
Hypoxia, a characteristic feature of many solid tumors, is known to play a crucial role in tumor progression and resistance to therapy. By targeting hypoxia-inducible factor 1 alpha (HIF1α), a key regulator of cellular response to low oxygen levels, the researchers aimed to develop a more effective strategy for tumor-specific drug delivery.
Previous efforts to target HIF1α have been hindered by the instability and limited binding abilities of the peptide-based molecules used. To overcome these obstacles, the team turned to MAPs, which offer the advantages of high stability and multivalency.
Drawing inspiration from their successful development of MAPs targeting COVID-19, the researchers engineered HIF1α-MAPs (H-MAPs) by grafting critical residues of HIF1α onto the MAP scaffold. This innovative design resulted in H-MAPs with picomolar binding affinities, significantly surpassing previous approaches. In vivo studies showed promising results, with H-MAPs effectively homing in on hypoxic tumors.
“This is a very promising result, using a material that is degradable within the body and likely will limit side effects to treatments,” said Montclare. “We’re taking advantage of the building blocks of our own bodies and using those protein compositions to treat the body — and that’s where we’re making big progress.”
Montclare’s findings suggest that H-MAPs hold great potential as targeted therapeutic agents for cancer treatment. With further refinement and exploration, H-MAPs could offer a new avenue for precision medicine, providing clinicians with a powerful tool to combat cancer while minimizing side effects and maximizing therapeutic efficacy.
The development of H-MAPs represents a significant advancement in the field of cancer therapy, highlighting the importance of innovative approaches that leverage the intricacies of the tumor microenvironment. As researchers continue to unravel the complexities of cancer biology, protein-based targeting agents like H-MAPs offer hope for improved outcomes and better quality of life for cancer patients.
Targeting stubborn breast cancer subtypes
Not all tumors are hypoxic, however, and some forms of cancers are much harder to target than others.
Triple-negative breast cancer (TNBC) poses a significant challenge in the realm of oncology due to its resistance to conventional targeted therapies. Unlike other breast cancer subtypes, TNBC lacks the receptor biomarkers — such as estrogen receptors and human epidermal growth factor receptor 2 — making it unresponsive to standard treatments. Consequently, chemotherapy remains the primary option for TNBC patients. However, the efficacy of chemotherapy is often hindered by the development of drug resistance, necessitating innovative approaches to enhance treatment outcomes.
In recent years, there has been a surge of interest in improving the efficacy of chemotherapy for TNBC through enhanced drug delivery systems. One promising avenue involves the use of biocompatible materials, including lipids, polymers, and proteins, as carriers to encapsulate chemotherapeutic agents. Among these materials, protein-based hydrogels have emerged as a particularly attractive option due to their biocompatibility, tunable properties, and ability to achieve controlled drug release.
A recent breakthrough in the field comes from the development of a novel protein-based hydrogel, known as Q8, which demonstrates remarkable improvements over previous materials
By fine-tuning the molecular characteristics of the hydrogel using a machine learning algorithm, the researchers were able to engineer Q8 to exhibit a two-fold increase in gelation rate and mechanical strength. These enhancements pave the way for Q8 to serve as a promising platform for sustained chemotherapeutic delivery.
In a groundbreaking study, the researchers investigated the therapeutic potential of Q8 for the treatment of TNBC in vivo using a mouse model. Remarkably, the delivery of doxorubicin encapsulated in Q8 resulted in significantly improved tumor suppression compared to conventional doxorubicin treatment alone. This achievement marks a significant milestone in the development of non-invasive and targeted therapies for TNBC, offering new hope for patients facing this aggressive form of breast cancer.
The success of Q8 underscores the immense potential of protein-based hydrogels as versatile platforms for drug delivery in cancer therapy. By leveraging the unique properties of these materials, researchers can overcome longstanding challenges associated with chemotherapy, including poor drug bioavailability and resistance. Moving forward, further advancements in protein engineering and hydrogel design hold the promise of revolutionizing cancer treatment paradigms, offering renewed optimism for patients battling TNBC and other challenging malignancies.
Unveiling biochemical defenses against chemical warfare
In the clandestine world of biochemical warfare, researchers are continuously seeking innovative strategies to counteract lethal agents. Researchers led by Jin Kim Montclare, Professor in the Department of Chemical and Biomolecular Engineering, have embarked on a pioneering mission to develop enzymatic defenses against chemical threats, as revealed in a recent study.
The team's focus lies in crafting enzymes capable of neutralizing notorious warfare agents such as VX, renowned for their swift and devastating effects on the nervous system. Through meticulous computational design, they harnessed the power of enzymes like phosphotesterase (PTE), traditionally adept at detoxifying organophosphates found in pesticides, to target VX agents.
The study utilized computational techniques to design a diverse library of PTE variants optimized for targeting lethal organophosphorus nerve agents. Leveraging advanced modeling software, such as Rosetta, the researchers meticulously crafted enzyme variants tailored to enhance efficacy against these formidable threats. When they tested these new enzyme versions in the lab, they found that three of them were much better at breaking down VX and VR. Their findings showcased the effectiveness of these engineered enzymes in neutralizing these chemicals.
A key problem in treating these agents lies in the urgency of application. In the event of exposure, rapid intervention becomes paramount. The research emphasizes potential applications, ranging from prophylactic measures to immediate administration upon exposure, underscoring the imperative for swift action to mitigate the agents' lethal effects.
Another key issue is protein stability — ensuring that the proteins can stay intact and at the site of affected tissue which is crucial for therapeutic applications. Ensuring enzymes remain stable within the body enhances their longevity and effectiveness, offering prolonged protection against chemical agents.
Looking ahead, Montclare's team aims to optimize enzyme stability and efficacy further, paving the way for practical applications in chemical defense and therapeutics. Their work represents a beacon of hope in the ongoing battle against chemical threats, promising safer and more effective strategies to safeguard lives.
Kronenberg, J., Chu, S., Olsen, A., Britton, D., Halvorsen, L., Guo, S., Lakshmi, A., Chen, J., Kulapurathazhe, M. J., Baker, C. A., Wadsworth, B. C., Van Acker, C. J., Lehman, J. G., Otto, T. C., Renfrew, P. D., Bonneau, R., & Montclare, J. K. (2024). Computational design of phosphotriesterase improves v‐agent degradation efficiency. ChemistryOpen. https://doi.org/10.1002/open.202300263