Laboratories and Centers
Tandon's biomedical engineers are building the technology for a healthier future. Our work straddles multiple areas of expertise, and spans a number of different fields and specializations.

Medical Imaging and Data Analysis
 This field involves designing new medical imaging instrumentation and methods as well as developing novel algorithms that help to analyze medical images and data. Artificial Intelligence and deep learning play an increasing role and our department collaborates closely with the Department of Radiology at NYU Langone.
This field involves designing new medical imaging instrumentation and methods as well as developing novel algorithms that help to analyze medical images and data. Artificial Intelligence and deep learning play an increasing role and our department collaborates closely with the Department of Radiology at NYU Langone.
Medical Imaging and Data Analysis Labs

Advanced Bio-Photonics and Imaging Laboratory
Professor Azhar Zam’s lab develops smart optical technologies for medical imaging, diagnostics, and monitoring. Their innovations include laser-based surgical systems, optical coherence tomography (OCT), photo-acoustics, biomedical spectroscopy, AI-assisted diagnostics, and miniaturized biosensors. By integrating advanced optics, AI, and miniaturized hardware, the lab enables detection and measurement of biological phenomena that are otherwise invisible—addressing critical unmet needs in biomedicine and clinical care.

Advanced MRI and Biomedical Imaging Laboratory
Professor Baete’s lab develops novel MRI techniques to improve the detection and monitoring of disease. Their innovations include advanced diffusion imaging and MRI pulse sequences that enhance brain microstructure mapping and cancer characterization. Collaborating closely with clinicians, the team translates these technologies into clinical practice, driving progress in neurological and oncological imaging for better patient outcomes.
Center for Advanced Imaging Innovation and Research
Professor Dan Sodickson’s research primarily addressed the development of new techniques for biomedical imaging to improve human health. He leads a multidisciplinary team that develops new methods for rapid continuous imaging, taking advantage of recent developments in parallel imaging, compressed sensing, and artificial intelligence. This work extends to clinical applications of MRI, PET and CT.
Clinical Artificial Intelligence Lab
Professor Farah Shamout leads the Clinical Artificial Intelligence (CAI) lab, where her team is developing machine learning models to support decision-making in healthcare. This includes AI approaches for the interpretation of large-scale multi-modal data, the prediction of in-hospital deterioration of CVOID-19 patients, and the assessment of breast cancer risk in women. In addition, she explores the fairness and ethics of AI.

Clinical Biophotonics Laboratory
Professor Andreas Hielscher’s team focuses on developing clinically relevant optical tomographic imaging systems. They apply these devices and wearable electronics to the diagnosis and treatment of various diseases, such as breast cancer, arthritis, peripheral artery disease, diabetic foot syndrome, and real-time monitoring of brain activities.

Computational Pathology and Precision Diagnostics Lab
Professor Tsirigos and his team develop AI-driven methods to analyze pathology images and advance cancer diagnostics. Their self-supervised learning framework uncovers novel tissue patterns without manual annotation, enabling more accurate prediction of clinical outcomes. By integrating histopathology with genomics and clinical data, the lab’s work enhances diagnostic precision and supports the development of personalized cancer therapies.
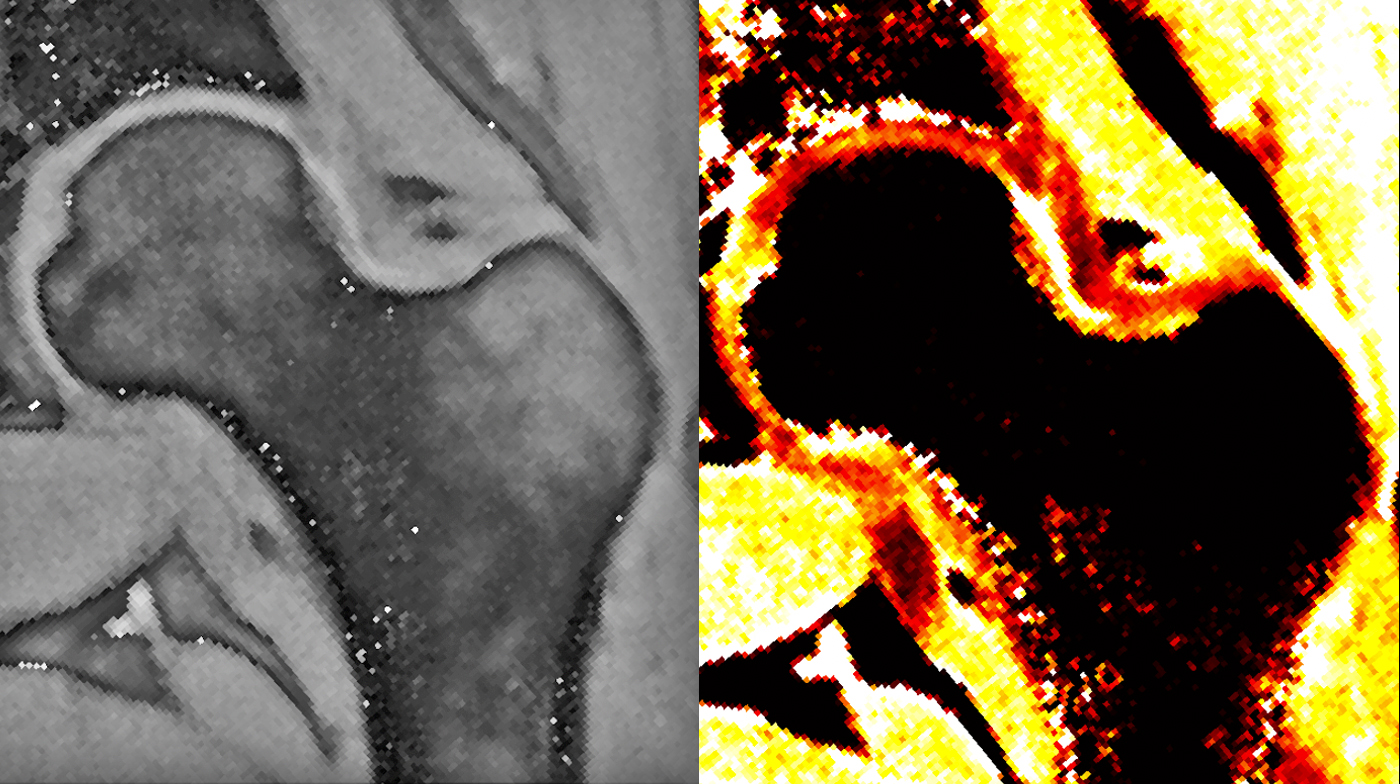
Lattanzi MRI Laboratory
Professor Lattanzi’s research lies at the boundaries between physics, engineering, and medicine. His group develops new techniques to improve the diagnostic power of magnetic resonance imaging (MRI). The main applications are in the areas of cartilage damage and hip osteoarthritis.
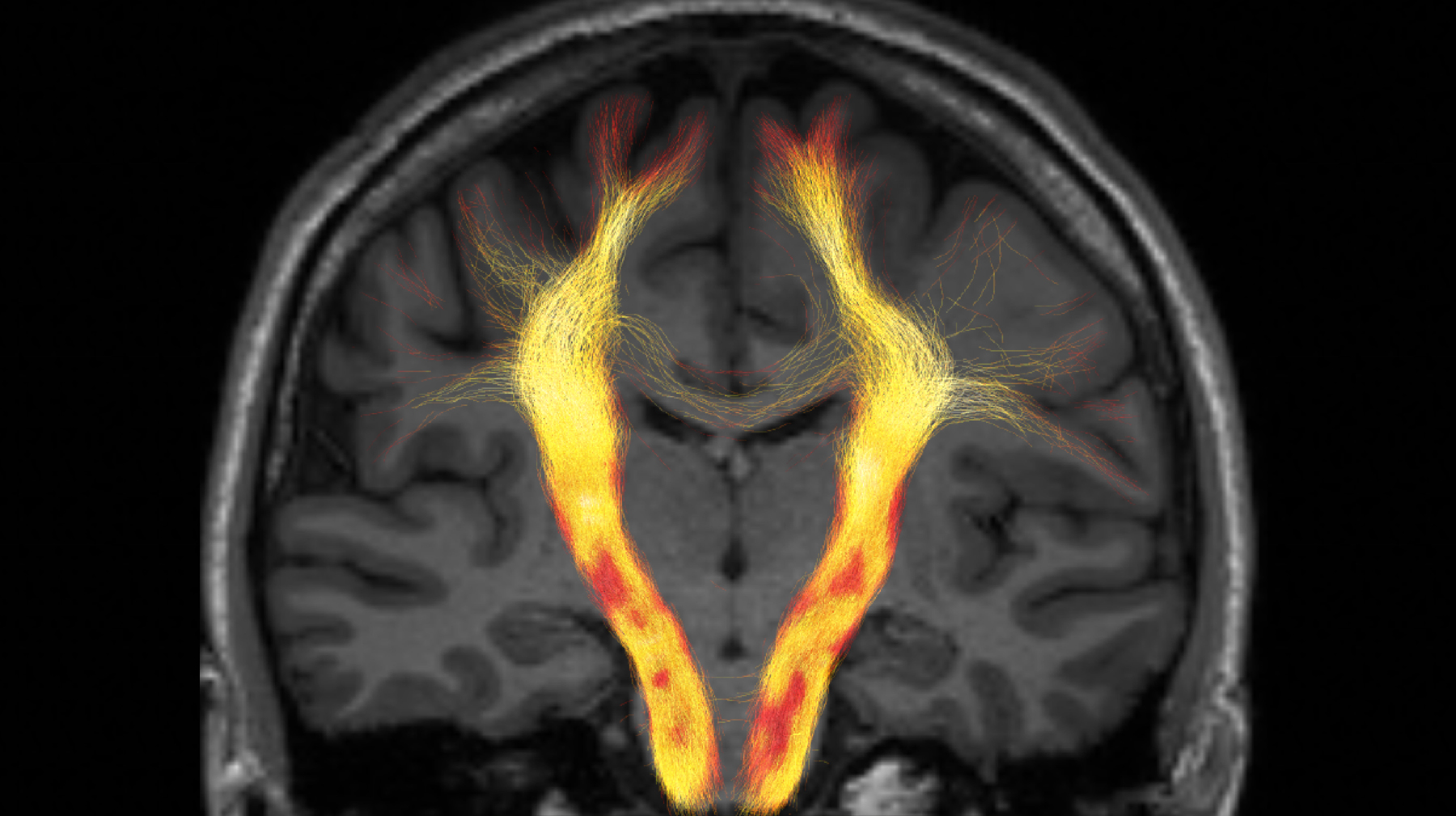
MRI Biophysics Group
Professor Els Fieremans works on validation methods for diffusion magnetic resonance imaging (MRI) including hardware, numerical phantoms, and animal models. Her group seeks to develop and translate new imaging markers for neurodegeneration (e.g. Alzheimer’s), as well as body diffusion applications in muscles and prostate cancer.
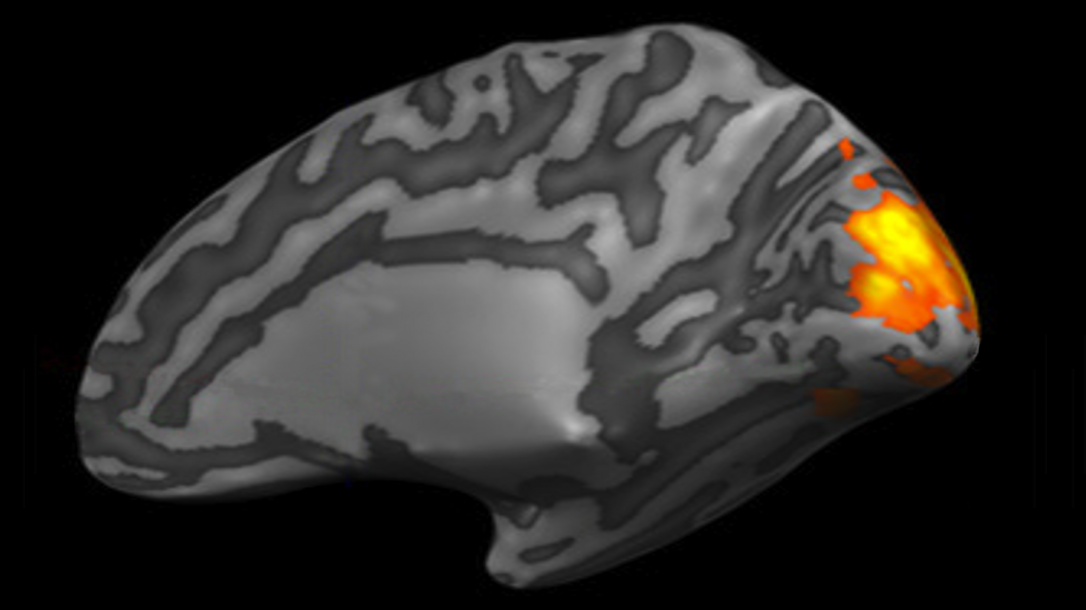
Neuroimaging and Visual Science Laboratory
Kevin Chan and his group are developing and applying new methods for noninvasive imaging of neurodegeneration, neurodevelopment, neuroplasticity, and neuroregeneration in people with vision-related diseases and injuries. They study the structural, metabolic, physiological, and functional relationships between the eyes, brain, and behavior with the mission to improve vision.
Neurophotonics Laboratory
Professor Srinivasan’s group at the Tech4Health Institute develops novel light-based technologies to image the structure and function of the eye and brain. The techniques include near-infrared spectroscopy, optical coherence tomography, and optical coherence microscopy. In close collaboration with clinicians, they improve the diagnosis and treatment of glaucoma and neurodegenerative diseases.
NYU Video Lab
Research activities in Professor Yao Wang’s group deal with encoding and distributing videos among a large number of users. Of special interest are diverse network access links and applications to biomedical imaging. The lab collaborates extensively with other research groups in wireless communication and networking at the School of Engineering and NYU’s medical school and hospitals.
Selesnick Lab
In the laboratory of Professor Ivan Selesnick, PhD, researchers are interested in digital signal processing, sparsity in signal processing, and multi-dimensional wavelet-based signal/image/video processing. They develop new methods for signal filtering, separation, and deconvolution, especially in the area of biomedical imaging.
Single Molecule Biophotonics Laboratory
Prof Eli Rothenberg’s team specializes in development of cutting-edge single-molecule fluorescence imaging techniques. They apply these techniques to studying molecular mechanisms relevant to diverse human diseases, including cancer. Adding novel computational methods and assays they aim to discover biomarker, therapeutic targets, and understand drug mechanisms-of-action.
Tissue Engineering and Regenerative Medicine
This area of biomedical engineering focuses on the assembly of functional constructs that restore, maintain, or improve damaged tissues or whole organs. It also incorporates research on self-healing — where the body uses its own systems, sometimes with the help of foreign biological material, to recreate cells and rebuild tissues and organs.
Tissue Engineering and Regenerative Medicine Labs
Bioprinting and Regenerative Design Lab
Professor Vijayavenkataraman’s lab integrates 3D printing, bioprinting, and computational modeling to engineer next-generation tissues, implants, and medical devices. The group designs biomimetic scaffolds for nerve, bone, and skin regeneration, develops sustainable bioinks from marine biomaterials, and fabricates conductive and vascularized tissue constructs to advance regenerative medicine and personalized healthcare.
Craniomaxillofacial Orthopedic Biomaterials Regenerative Applications Lab
Professor Lukasz Witek’s research group performs in vivo and in vitro evaluations of a wide range of (bio)materials that can be used as implants for dental and orthopedic applications. A special focus is on 3D printing of bio-ceramic materials for tissue regeneration.
Kirsch Lab
The ultimate goal of Professor Kirsch’s research is to define novel therapeutic targets for the treatment of osteoarthritis and other joint diseases and injuries. To this end the laboratory studies mechanisms involved in the regulation of cartilage homeostasis, maintenance and pathology. In addition, research is performed on how cells and the joint environment interact.

Laboratory of Human Organogenesis
Professor Rosado-Olivieri’s lab explores the cellular and molecular mechanisms driving human organ development. They develop innovative stem cell-based mini-organ models and computational tools to study tissue morphogenesis and differentiation. By uncovering how these processes are disrupted in disease, the team aims to advance regenerative therapies and improve disease modeling for clinical applications.
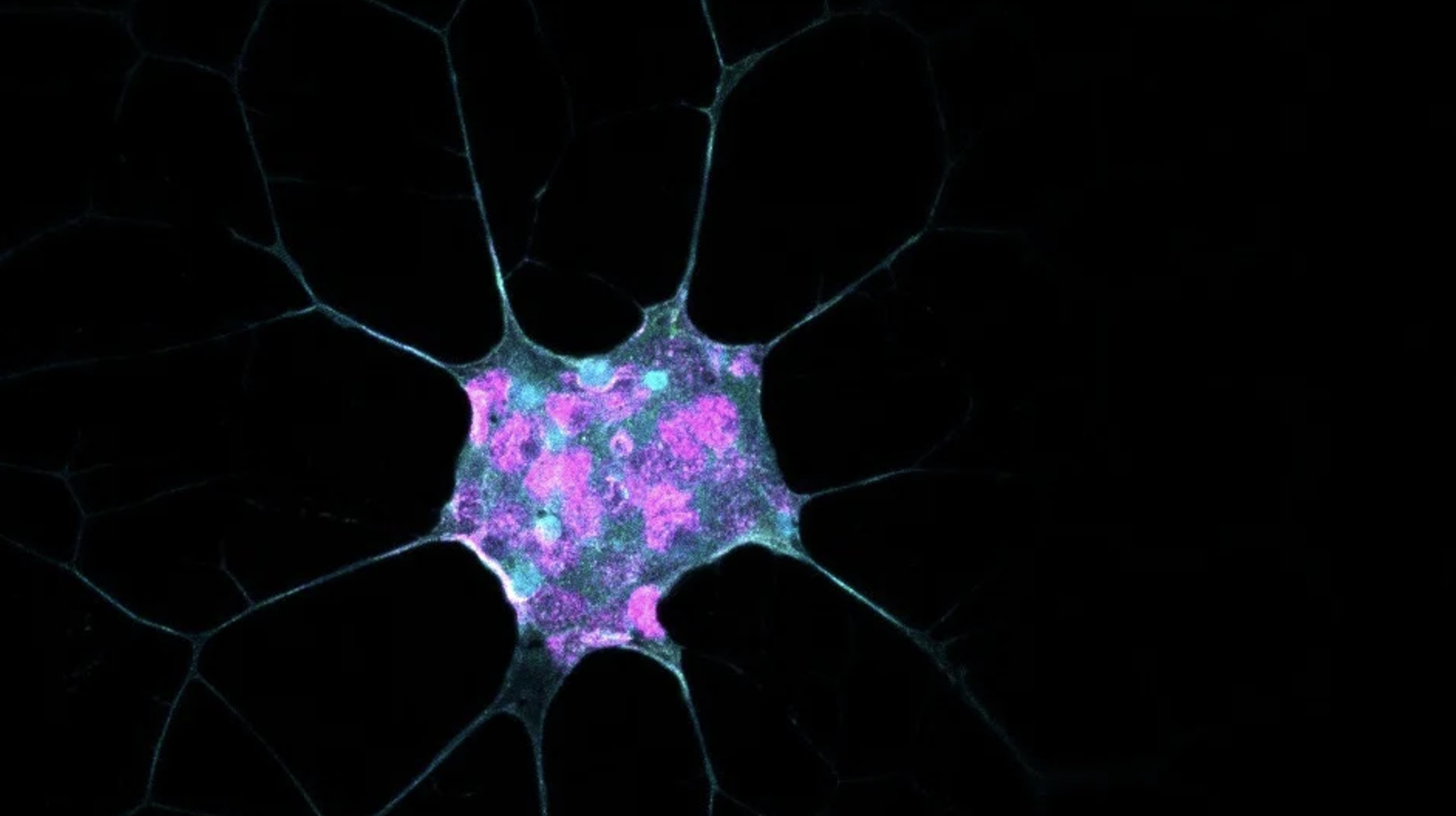
NanoBioEngineering for Tissue Regeneration Laboratory
Professor de Lázaro is interested in understanding how genetic, biochemical, and mechanical signals impact cell plasticity, identity, and function. The lab’s mission is to apply this knowledge in combination with nanomedicine and bioengineering strategies to develop therapeutic interventions. A major focus lies on immunotherapies and tissue regeneration/rejuvenation for heart diseases.
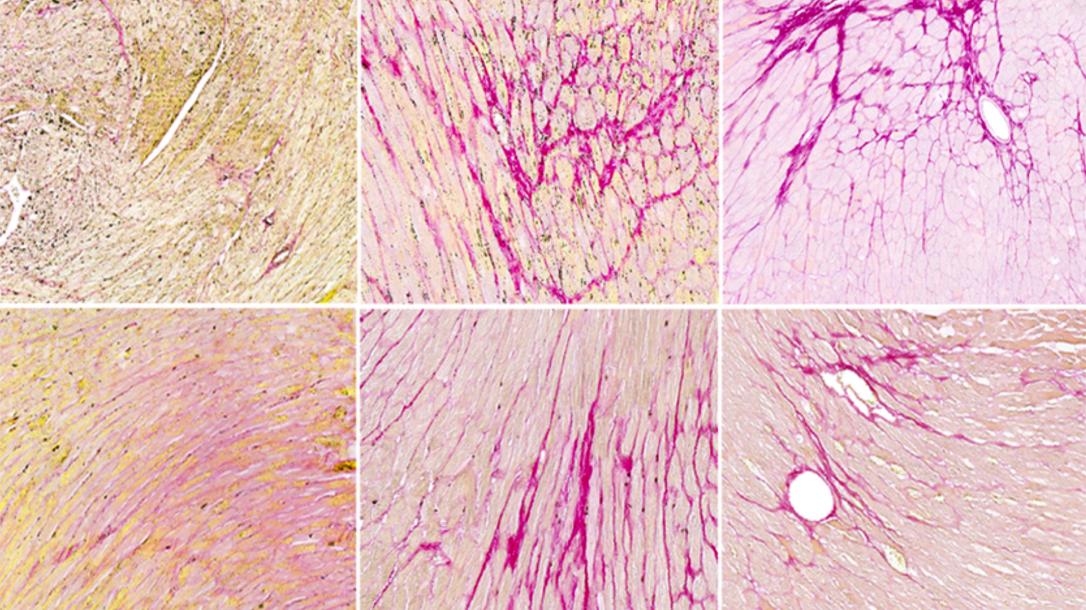
Shukti Chakravarti Lab
Professor Chakravarti’s research group tries to understand how the extracellular matrix (ECM) regulates cellular functions and tissue homeostasis. In their work they use cell cultures and mouse models to gain a better understanding of the role of neutrophil, macrophage and dendritic cell functions in bacterial and viral infections. In addition, they explore the ECM changes and the underlying genetic causes in corneal diseases.
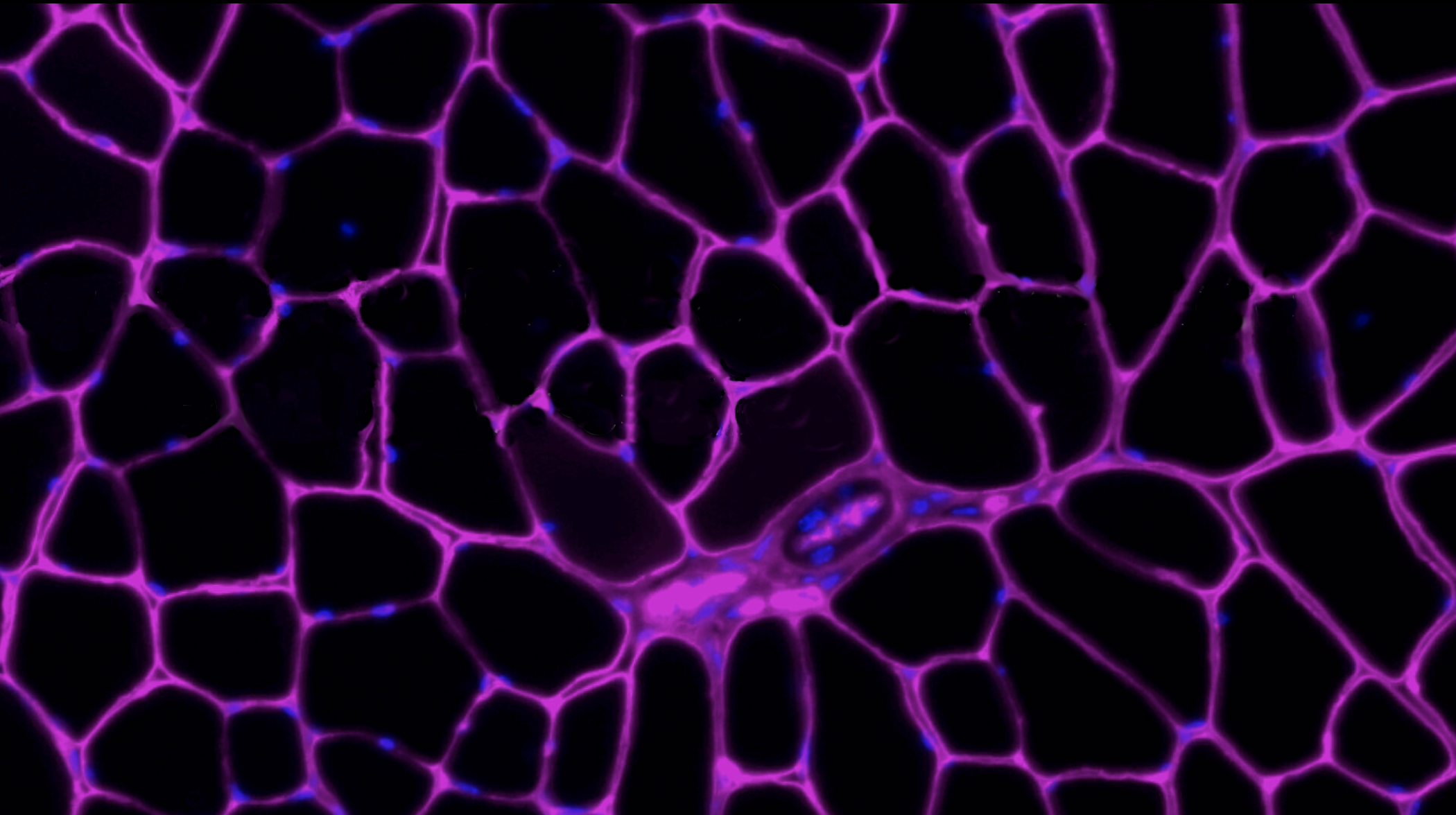
Tissue Maintenance and Regeneration Laboratory
Tissue maintenance and regeneration are crucial for preserving tissue functionality and organismal health. With age and disease, both of these processes can become inefficient. Professor Wosczyna’s laboratory seeks to identify cellular mechanisms that are responsible for pathological phenotypes. They look for molecular targets that can be modified to extend tissue functionality further into age.
Synthetic and Systems Bioengineering
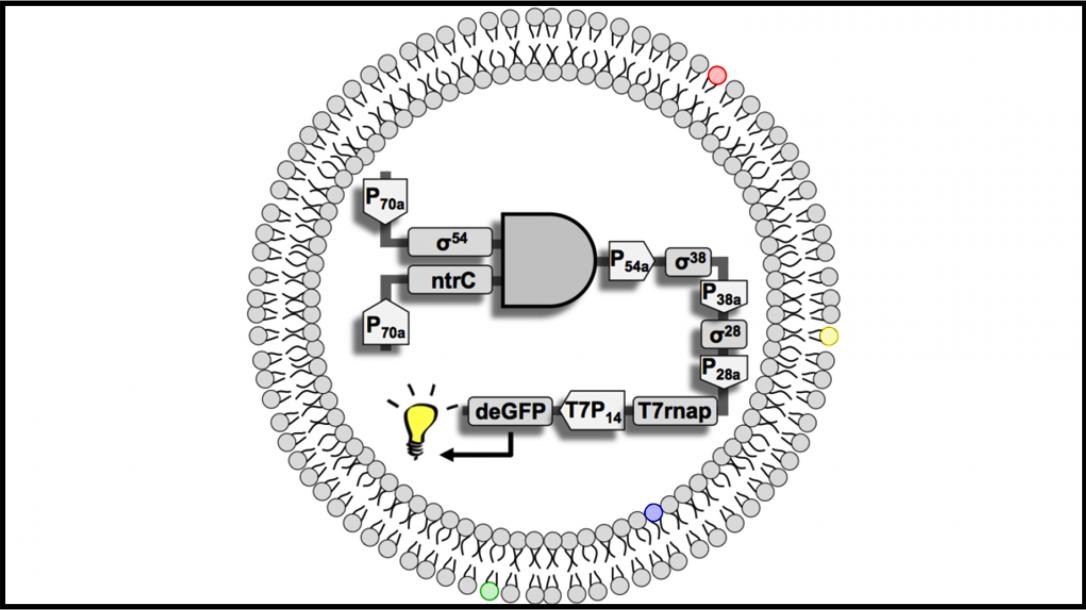 This relatively young field is based on the premise that living systems are modular, and thus, able to be engineered. The emphasis lies on designing genetically encoded information and cell micro environments to gain a deeper understanding of living things as well as to generate useful products, such as advanced medical treatments.
This relatively young field is based on the premise that living systems are modular, and thus, able to be engineered. The emphasis lies on designing genetically encoded information and cell micro environments to gain a deeper understanding of living things as well as to generate useful products, such as advanced medical treatments.
Synthetic and Systems Bioengineering Labs
Boeke Lab
The laboratory of Professor Jef Boeke is well known for foundational work on mechanistic and genomic aspects of retro-transposition in both yeast and mammalian systems. After more than three decades, they continue to scrutinize their favorite genomic parasites. In addition, the Boeke lab is heavily involved in the development of novel technologies in genetics, genomics and synthetic biology.
Programmable Regenerative Immunity Lab
The Programmable Regenerative Immunity Lab, led by David Truong, builds universal human cell platforms for responding to disease with precision. We use synthetic biology, genome engineering, and iPSC engineering to design programmable immune and neural cells — such as dendritic cells, macrophages, and thymic epithelial cells. These systems are programmed to interpret disease-specific signals and restore physiological function in cancer, neurodegeneration, and immune dysfunction. Our long-term goal is to enable context-aware living therapies that reprogram how the body heals itself.

Dynamical Systems Laboratory (DSL)
Professor Maurizio Porfiri’s group conducts multidisciplinary research in the theory and application of dynamical systems, motivated by the objectives of advancing engineering science and improving society. They perform research and development in the areas of robotics and mechatronics, experimental fluid mechanics, material characterization, animal behavior, and vibrations.
Laboratory of Computational Multiomics
Professor Ruggles’ laboratory focuses on understanding human health and biology using data science, data visualization, and predictive modeling. A primary goal of this research is to analyze and integrate diverse data modalities, including bulk and single-cell sequencing, phospho- and global- proteomics, metagenomics, flow cytometry, imaging, and clinical data. These multi-omic methods are used to better understand cancer, heart diseases, and other disorders.
Laboratory of Computational Proteomics
Prof. David Fenyö’s uses proteomic approaches to develop methods to identify, characterize, and quantify proteins important for various cellular processes. His efforts to integrate data from multiple technologies - including mass spectrometry, sequencing, and microscopy - have provided a wide array of powerful tools to discover and verify biomarkers and therapeutic targets in cancer.
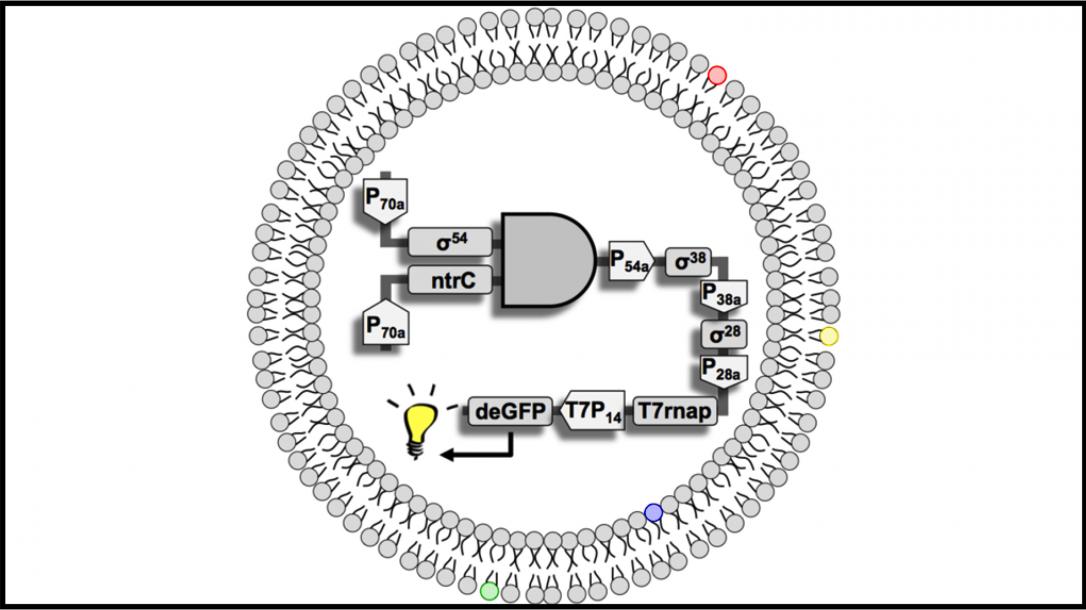
Quantitative Synthetic Biology Lab
Professor Andras Gyorgy’s research focuses on networked dynamical systems. In particular, he is interested in rational forward-engineering of synthetic biology systems; characterization of circuit-circuit and host-circuit interactions both in vivo and in vitro; and the design and analysis of multi-scale systems prevalent in biology.
Rational Microbiome Engineering Lab
Professor Jonas Schluter’s goal is to turn the human microbiome into a therapeutic target for completely novel medical therapies. To this end, his team is using machine learning, AI, and Bayesian inference to analyze massive clinical data sets and electronic health records. They also perform lab experiments and clinical studies to understand how the microbial ecosystem works and can be changed.
Robotic and Rehabilitation Engineering
 The main goals are the development of technological solutions and devices to assist individuals with disabilities and aid the recovery of physical and cognitive functions lost because of disease or injury. New technologies, including advanced brain-machine interfaces, increase mobility, communication, hearing, vision and cognition of affected patients.
The main goals are the development of technological solutions and devices to assist individuals with disabilities and aid the recovery of physical and cognitive functions lost because of disease or injury. New technologies, including advanced brain-machine interfaces, increase mobility, communication, hearing, vision and cognition of affected patients.
Rehabilitation Engineering Labs

Biomechatronics and Intelligent Robotics Lab
The Biomechatronics and Intelligent Robotics Lab's research focuses on innovative robotic technologies to assist people in areas like walking, surgery, and rehabilitation. Sample projects include a reinforcement learning-powered exoskeleton that reduces human energy use, a versatile catheter robot for safer surgeries, and MRI-guided robots to improve prostate cancer biopsies

Physical Therapy and Exercise Lab
Professor Smita Rao and her colleagues in the Center of Health and Rehabilitation Research study the effects of exercise in individuals with diabetes, neuropathy, osteoarthritis, and other musculoskeletal conditions. Her research focuses on improving physical therapy and rehabilitation care in these patients.

Rusk Rehabilitation
Advances in miniaturized sensors and actuators, as well as artificial intelligence (AI), have broadened horizons for assistive and rehabilitative technologies. The team of Professor JohnRoss Rizzo, MD, is leveraging these innovations to help patients with conditions such as blindness and stroke, enhancing their ability to interact physically with their environment.
Nano- and Micro- Bioengineering
 This branch of engineering involves the development and application of materials and devices to study biological processes and to treat disease at the level of single cells, molecules and atoms. A particular strength at NYU are bio-fluidics and lab-on-a-chip technologies.
This branch of engineering involves the development and application of materials and devices to study biological processes and to treat disease at the level of single cells, molecules and atoms. A particular strength at NYU are bio-fluidics and lab-on-a-chip technologies.
Nano- and Micro- Bioengineering
Advanced Microfluidics and Microdevices Laboratory
Professor Qasaimeh’s group is developing micro and nanotechnologies for healthcare applications. Of special interest are devices for point-of-care diagnostics that use blood samples to detect, for example, metastatic tumor cells or fetal cells for genetic analysis. His research elucidates how cancer cells migrate in the body and how white blood cells respond to infections.
Applied Micro-Bioengineering Laboratory (AMBL)
Taking advantage of state-of-art nanotechnologies and fabricating fascinating functional biomaterials and integrated biosystems, Professor Weiqiang Chen’s laboratory addresses numerous important problems in fundamental biology as well as clinical applications in disease diagnosis and treatment.

Integrated BioElectronics Laboratory
Professor Sohmyung Ha’s lab aims at advancing the engineering and applications of silicon integrated technology interfacing with biology. This is approached in a variety of forms, ranging from implantable biomedical devices to unobtrusive wearable sensors.

Micro- and Nano-scale Bioengineering Lab
The research in the Professor Yong-Ak (Rafael) Song’s group at NYU-AD lies at the interface between biology, physics and engineering. More specifically, their research is focused on applying microfluidics and nanofluidics to broad range of challenges in bioscience and medicine.
MicroParticle PhotoPhysics Laboratory for BioPhotonics
We are involved in inventing cutting-edge photonic techniques for detecting individual virus particles. We recently detected single Influenza A virus in-vitro and is pushing its patented Whispering Gallery Mode Biosensor to higher sensitivity in order to detect single HIV virions.
Nanooptical Bioengineering Lab
Hoagang Cai’s lab advancec nanotechnology for biological and biomedical applications. Employing nanolithographic technology, his group creates designer biomaterials and metasurfaces with unprecedented precision and functionality. Applications range from studying T-cell activation in cancer treatment to photo-stimulation in optogenetics and brain research.
Pinkerton Nanomaterials Research Group
Nanomedicine is a fast-emerging field that focuses on the development and application of nanomaterials to diagnose and treat diseases. Professor Nathalie Pinkerton’s team focuses on the creation of functional soft materials via scalable synthetic processes. To that end they employ tools from chemical and materials engineering, nanotechnology, chemistry, and biology. Their work leads to a better understanding of nanomaterial behavior in biological systems and, for example, more sensitive contrast agents for medical imaging.
Protein Engineering and Design Lab
Jin Kim Montclare’s lab is performing groundbreaking research in engineering proteins to mimic nature and, in some cases, work better than nature. Her team works to customize artificial proteins with the aim of targeting human disorders, drug delivery and tissue regeneration as well as create nanomaterials for electronics. Applications of this multidisciplinary research in chemistry and genetic engineering range from new vehicles for drug delivery to treating breast cancer.

Reverse Bioengineering Laboratory
The primary objective of Professor Kenichiro Kamei’s lab is to revolutionize the field of biomedical engineering by reconstructing complete human and animal bodies using cutting-edge engineering techniques. A prime example of their work is the development of the “Body on a Chip” (BoC) system, which simulates physiological and pathological conditions of living organisms in vitro. This work has far-reaching implications for drug discovery and regenerative medicine.
Neuroengineering
 Neuroengineering is a multidisciplinary field that involves the use of engineering technology to study and influence neurodegenerative diseases and function of the brain. Research at NYU is primarily focused on interfacing with the nervous system either by novel non-invasive imaging methods or the development of implantable technology and materials.
Neuroengineering is a multidisciplinary field that involves the use of engineering technology to study and influence neurodegenerative diseases and function of the brain. Research at NYU is primarily focused on interfacing with the nervous system either by novel non-invasive imaging methods or the development of implantable technology and materials.
Neuroengineering Labs
Angelaki Lab
The systems and computational neuroscience laboratory of Professor Dora Angelaki focuses on how the brain generates perception and cognition. Her group studies how multi-sensory signals flow dynamically across brain areas, how causal inference is implemented in the brain, how beliefs propagate through the network, and how internal states modulate this information flow.

Artificial and Biological Computation Laboratory
Professor Cristina Savin investigates how neural circuits represent, update, and adapt beliefs to manage uncertainty and change. By combining machine learning, probabilistic modeling, and experimental neuroscience, they explore the computational principles underlying learning, memory, and decision-making. Their work bridges biological and artificial systems, offering insights into how adaptable cognitive processes are implemented in both the brain and engineered networks.

Cancer Pain and Neurobiology Laboratory
The team of Professor Yi Ye investigates how peripheral nerves interact with the tumor microenvironment to drive both oral cancer progression and pain. Using advanced imaging, electrophysiology, genetic tools, and behavioral models, they uncover shared mechanisms underlying cancer growth and pain signaling. Their work aims to inform the development of novel therapies that target both cancer and cancer-associated pain.

Center for Dementia Research
Professor Ginsberg’s group investigates how neurons adapt at the molecular and cellular levels following brain injury and in neurodegenerative diseases. Focusing on the hippocampus, they analyze single neurons to uncover early markers of synaptic and dendritic reorganization. Using advanced RNA and proteomic techniques, their work provides insights into mechanisms of neuroplasticity and the progression of Alzheimer’s disease and related disorders.

Computational Neuroscience, Neuroengineering, and Neuropsychiatry Laboratory (CN3)
The research in Professor Sage Chen’s lab aims to find solutions for real-time brain-machine interfaces, pain management, and psychiatric disorders. Developing a better understanding of memory and sleep is another goal. To accomplish all this, they advance and integrate the fields of computational neuroscience, neural engineering, and machine learning.

Laboratory for Advanced Neuroengineering and Translational Medicine
Professor Khalil Ramadi and his team develop innovative approaches for the modulation of neural activity throughout the body. The goal is to come up with novel therapies for neurologic, metabolic, and immune disorders. They combine mechanical, electrical, materials, and bio-engineering toolkits in the design of minimally invasive technologies.

Laboratory for Comparative Brain Dynamics
Despite their centrality to everyday life, the neural underpinnings of motor sequences in human speech are poorly understood. Prof. Long’s group seeks to identify the relevant brain processing centers involved in communication. To accomplish this, they focus on three distinct types of behaviors and models: song production in the zebra finch, counter-singing in Alston’s brown mice, and human speech.

Medical Cyber-Physical Systems Laboratory
Professor Rose Faghih group develops biomedical signal processing and control algorithms for human-technology interactions and monitoring. These state-of-the-art tools are employed for prognosis, diagnosis, and treatment of pathological conditions related to neuro-endocrine and neuro-psychiatric disorders.
Neural Circuits and Algorithm Group
The aim of Dr. Chklovskii’s research is to understand how the brain analyzes large and complex datasets streamed by sensory organs. Informed by anatomical and physiological neuroscience data, his group develops algorithms that model brain computation and solve machine learning tasks. The overarching goal it to build artificial neural systems and treating mental illness.
Neural Interface Engineering Lab
The team of Professor Shy Shoham works at the interface of neuroscience and engineering, developing and applying modern bidirectional neural interfaces for observing and controlling neural population activity patterns. Their goal is to better understand sensory-motor information coding and to advance medical neurotechnology.
Pain Management Laboratory
The human experience of pain is incredibly complex. Understanding how pain affects patients, and how best to manage it, is Prof. Jing Wang’s goal. His research interest is centered on the role of brain circuits in the regulation of acute and chronic pain. Of particular interest are the cortical mechanisms of pain processing and regulation
Perception and Brain Dynamics Laboratory
Every day, our brains cycle through different states of awareness: from the rich conscious experiences during wakefulness to dreamless sleep to the bizarre experiences during dreaming sleep. Understanding the neural basis of conscious awareness is the basic goal of Prof. Biyu He’s team. She is using a combination functional magnetic resonance imaging (fMRI), magneto- encephalography (MEG) and invasive electrophysiology to explore relevant neural mechanisms and characterize various neuropsychiatric illnesses.
Schultebraucks Computational Psychiatry Lab
Professor Katharina Schultebraucks applies advanced computational methods to improve individualized risk prediction and treatment allocation in mental health. Her research integrates behavioral, neuroendocrine, molecular, and genetic data from longitudinal studies to identify early predictors of disorders such as post-traumatic stress disorder (PTSD). With a focus on precision psychiatry, her lab bridges digital phenotyping, machine learning, and translational neuroscience to advance personalized approaches to psychiatric care.

Social Behavior Neuroscience Lab
Social behaviors such as fighting, defense, and parenting, are innate and ubiquitous across the animal kingdoms. The research in Dr. Lin’s laboratory centers on understanding the neural circuits underlying these behaviors. Various genetic engineering, tracing, functional manipulation, in vivo electrophysiological recording and computational tools are combined to dissect the neural circuits in a great detail.
Speech Language Electrocortiography Laboratory
How is language produced and perceived in the human brain? What are the network dynamics that allow us to fluently communicate? These questions are still poorly understood and collaborations between scientists, clinicians, and engineers are crucial for making progress in this fascinating field. Adeen Flinker and his team are using unique human neurosurgical recordings and advanced data processing algorithms to elucidate these questions.
Cell and Immuno Bioengineering
 This emerging area involves the application of engineering tools and principles to quantitatively study the immune system in health and diseases. The overarching goal is the development of therapeutic interventions for precisely controlling and modulating a patient's immune response, e.g. to fight cancer.
This emerging area involves the application of engineering tools and principles to quantitatively study the immune system in health and diseases. The overarching goal is the development of therapeutic interventions for precisely controlling and modulating a patient's immune response, e.g. to fight cancer.
Cell and Immune Bioengineering Labs

Cardiovascular Disease and Lipid Metabolism Lab
Professor Edward Fisher’s lab studies the molecular mechanisms that drive cardiovascular disease. They investigate how liver lipoprotein production contributes to blood lipid levels and fatty liver disease. The team also explores how cholesterol-laden plaques form and regress in arteries, identifying strategies that combine lipid lowering with inflammation resolution. Their goal is to develop new therapies for atherosclerosis and related cardiometabolic conditions.

Davoli Lab
What are the causes and consequences of genomic copy number alterations (i.e. aneuploidy) in cancer and how does this affect patients’ response to therapy, especially immuno-therapy? These are among the main questions Prof. Teresa Davoli’s lab is interested in. To answer these questions, they utilize a variety of experimental and computational approaches, from large-scale genetic screens in human cells, to prediction of survival in cancer patients.
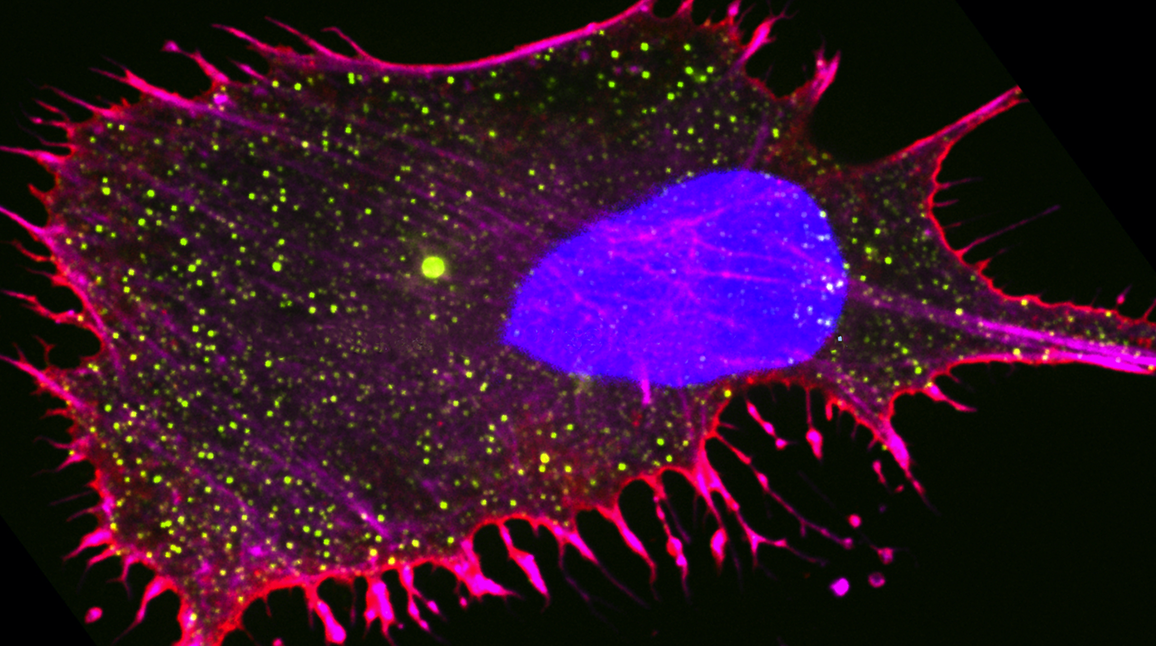
Holt Lab
Professor Liam Holt and his team are investigating how perturbations to the physical properties of the cell interior through mechanical forces impact cell and developmental biology. They apply this knowledge to improve ways to treat cancer and neurodegenerative diseases, such as Alzheimer’s.

Laboratory for Immuno Bioengineering Research and Applications
Professor Jeremy Teo and his LIBRA team study the mechanobiology of single immune cells and the implications of microenvironmental cues in down-stream immune biology. They are interested in depicting the mechanisms of these signals and modulating the immune outcome using bioengineering strategies. The overall goal is the development of technologies for translational therapeutics.
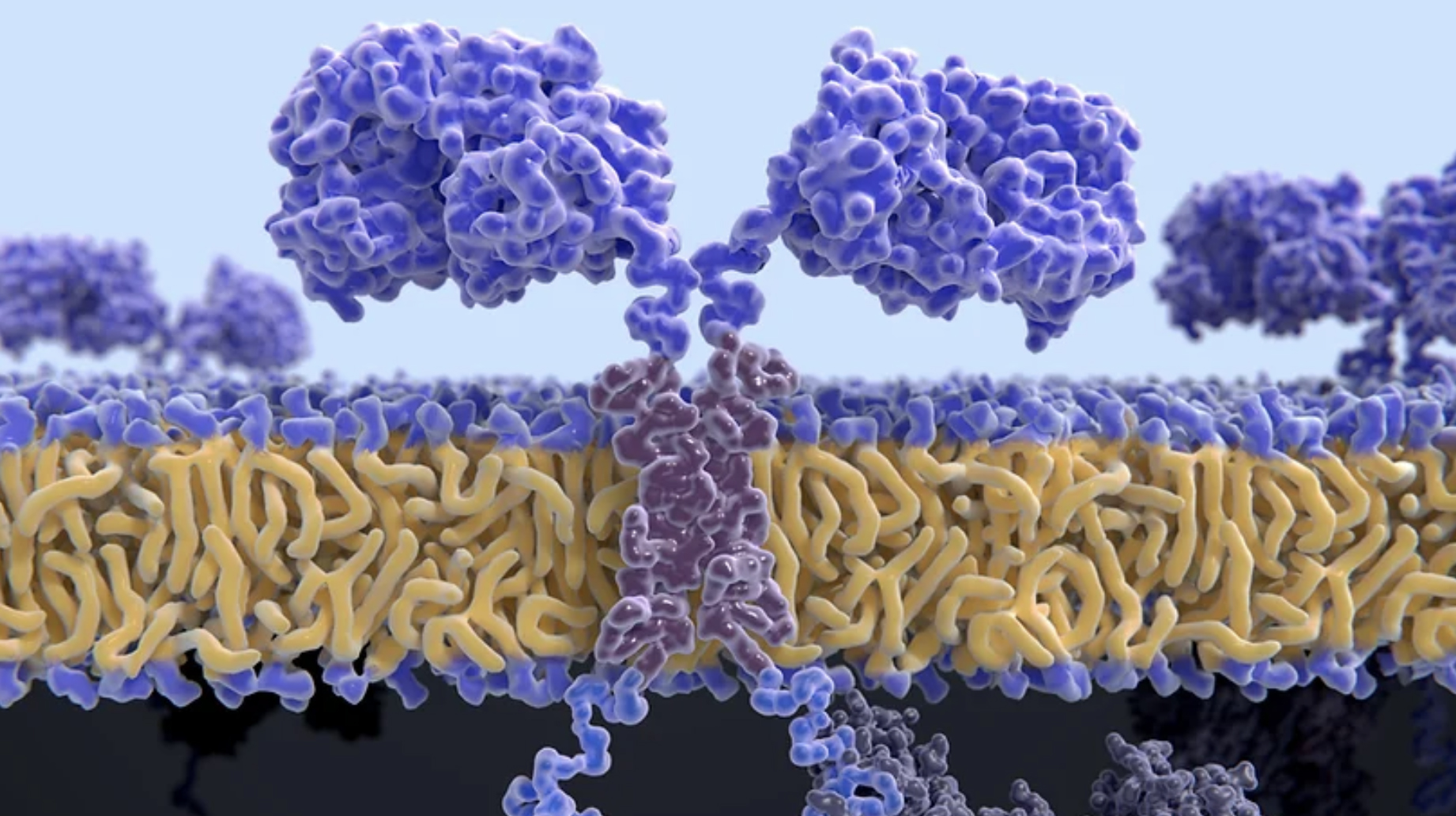
Laboratory for Personalized Immunotherapies
Personalized immunotherapies involving CAR T cells, TCRs and BiTEs, have revolutionized the treatment of cancer. They provide in cures in subsets of cancer patients, yet remain ineffective for the majority of patients. Prof. Mark Yarmarkovich aims to expand the use of curative immunotherapies. To this end, his group develops and employ technologies at the intersection of genomics, proteomics, immunology, antibody engineering and computational biology.
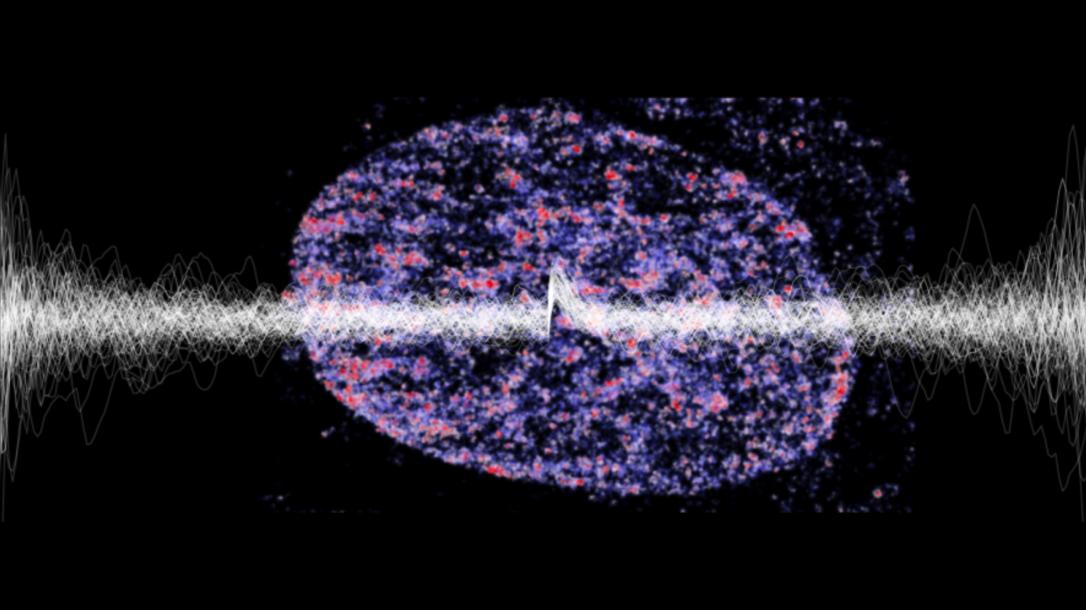
Lionnet Lab
To respond to a changing environment, cells have to express the right genes at the right time. Prof. Timothy Lionnet’s lab tries to understand how exactly this is achieved and how robust responses emerge from random molecular events. Getting insights into these biomolecular processes will ultimately provide new tools to tackle, for example, wound healing, cancer, and many other diseases.

Tumor Immunology and Immunotherapy Lab
Professor Jun Wang’s lab develops next-generation immunotherapies that go beyond PD-1/PD-L1 blockade. By integrating protein engineering, drug design, and disease modeling, they aim to enhance tumor-specific immunity while minimizing side effects. Their work identifies novel therapeutic targets and biomarkers to improve outcomes in cancer, autoimmune, and infectious diseases - advancing personalized and more effective immunotherapy strategies.