The Path from “Eureka!” to a Living Drug
Speaker:
Mark Tomishima, Ph.D.
Senior Vice President
BlueRock Therapeutics
Abstract:
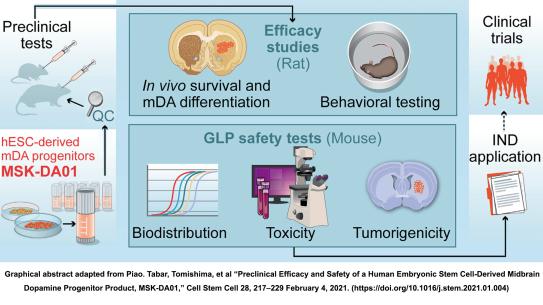
Transforming a scientific breakthrough into a therapeutic that can be delivered safely to patients is a long and complex journey, especially when the “drug” is a living cell. This talk will trace the path from early discovery in pluripotent stem cell (PSC) biology to the development of clinically manufactured cell therapies. Dr. Tomishima will discuss how advances in genome engineering, including ZFNs, TALENs, and CRISPR/Cas, enabled precise modification of human PSCs for purification, incorporation of safety switches, reporter integration, and enhanced functional performance. He will examine how engineered cell lines are designed to meet stringent criteria for identity, purity, potency, genetic stability, and reproducibility, and how differentiation protocols must be optimized for scalability and regulatory compliance. A central focus will be the translational gap between academic proof-of-concept studies and first-in-human trials. Dr. Tomishima will describe how research-grade differentiation methods are converted into closed-system, Good Manufacturing Practice (GMP)-compliant processes, including raw material qualification, in-process controls, release testing, batch record development, and comparability assessments. He will also address analytical assay development, cryopreservation strategies, supply chain considerations, and long-term product characterization needed to ensure consistent clinical performance. Using the example of , BlueRock Therapeutics’ lead cell therapy candidate, he will illustrate how multidisciplinary collaboration between academia, biotechnology, and GMP manufacturing facilities converts an early discovery into a viable clinical product. He will further discuss how strategic industry partnerships and integrated cell and gene platforms expand the therapeutic potential of PSC-derived products. The talk will highlight not only scientific innovation, but also the operational, regulatory, and strategic decisions required to translate stem cell engineering into a living drug.
Dr. Mark Tomishima started his career with a B.S. in Biology at San Diego State University and worked as a research technician at The Scripps Research Institute. Subsequently, he earned his Ph.D. in Molecular Biology at Princeton University. He then joined the laboratory of Lorenz Studer as a postdoctoral fellow at Memorial Sloan Kettering Cancer Center (MSKCC), where he began focusing on pluripotent stem cell biology and on genetic engineering approaches for cell-replacement strategies. He later founded and directed the SKI Stem Cell Research Facility at MSKCC, building it into a nationally recognized resource that supported investigators across institutions, trained more than 140 scientists, and helped establish best practices for pluripotent stem cell culture and translation. In 2017, Dr. Tomishima transitioned to BlueRock Therapeutics, where he has held successive leadership roles spanning genome engineering, CNS bioprocessing, and platform discovery, and now serves as Senior Vice President. His career reflects a rare continuum, from grad student to core facility builder to bio-tech executive, shaping the field of regenerative medicine across academia and industry.