Miguel Modestino
-
Director, Sustainable Engineering Initiative
-
Donald F. Othmer Associate Professor of Chemical Engineering

Our research lies at the interface of multifunctional material development and electrochemical engineering. Electrochemical devices are ubiquitous to a broad range of energy conversion technologies and chemical processes. Their core components rely on complex materials that provide the required electrocatalytic activity and mass transport functionality. Our group has expertise in composite materials development, processing and characterization; and this expertise is used to improve and redefine electrochemical reactors with direct industrial applications. Our applied research approach also relies on fundamental understanding of the materials’ self-assembly and how their morphology and surface properties affects the mass transport and performance of electrochemical devices.
Education
University of California, Berkeley 2013
Ph.D., Chemical Engineering
Massachusetts Institute of Technology 2008
M.S. , Chemical Engineering Practice
Massachusetts Institute of Technology 2007
B.S., Chemical Engineering
Awards
- Goddard Junior Faculty Fellowship Award, NYU (2020)
- MIT Technology Review Innovators Under 35 Award, Global List (2020)
- National Science Foundation CAREER Award (2019)
- Doctoral New Investigator Award, ACS Petroleum Research Fund (2018)
- MIT Technology Review Innovators Under 35 Award, Latin America (2017)
- Global Change Award, H&M Foundation (2017)
- Energy and Environmental Science Readers’ Choice Lectureship award (2015)
Research News
Bubble Trouble: New Research Highlights Outsized Impacts of Tiny Bubbles in Water Electrolysis
Hydrogen is often described as the fuel of the future — a clean, energy-dense way to store renewable power and decarbonize industries from steelmaking to shipping. But inside the devices that produce it, a surprisingly small and familiar phenomenon is getting in the way: bubbles.
In water electrolysis, electricity splits water into hydrogen and oxygen gases. Those gases naturally form bubbles on the surfaces of electrodes. For decades, researchers have focused on improving catalysts and materials to make this process more efficient. Yet a new paper by postdoctoral researcher Darjan Podbevšek and Sustainable Engineering Initiative Director and Associate Professor of Chemical and Biomolecular Engineering Miguel A. Modestino published in the journal Joule argues that the real bottleneck may be far more mundane.
“Bubble dynamics represent a largely overlooked bottleneck that can account for significant efficiency losses.” says Podbevšek.
At first glance, bubbles might seem harmless, even expected. But their presence sets off a cascade of problems. As bubbles stick to electrode surfaces, they block the very sites where reactions are supposed to occur. They also disrupt the flow of charged particles in the liquid, increasing electrical resistance. As more bubbles accumulate, they can create uneven conditions across the electrode, further degrading performance.
In some cases, these effects are not trivial. The authors note that bubble-related losses can range from about 5 percent to as much as 25 percent of the total energy input, depending on operating conditions. In a technology where efficiency directly affects cost and scalability, that’s a major obstacle.
What makes the problem especially challenging is that bubbles behave in complex ways across multiple scales. At the smallest level, they begin as tiny nuclei, often forming at microscopic imperfections on the electrode surface. Their growth depends on subtle forces, including surface tension and gradients in temperature or concentration.
As bubbles grow and detach, they interact with one another, merging into larger bubbles or forming tiny bubble layers that blanket the electrode. These microscale “bubble carpets” can fundamentally alter electrode/electrolyte interactions and how reactants and products are transported in the system.
“It’s how and where they evolve, grow, detach, and interact that determines their impact,” the authors emphasize.
This multiscale complexity helps explain why the problem has been so difficult to tackle. Many experiments focus on single bubbles under highly controlled conditions, but real electrolyzers operate in messy, turbulent environments filled with countless interacting bubbles. Bridging that gap, the authors argue, is essential for making meaningful progress.
A typical electrolyzer is a tightly sealed, windowless box, operating at high pressure and temperatures (>30bar, >80°C), with highly alkaline or acidic conditions, making it difficult to observe bubbles directly. The authors suggest that it is at least part of the reason there are comparatively few bubble-related studies in the field.
The emerging view is that bubbles are not just a nuisance but an engineering challenge — one that can be managed, and perhaps even exploited. Researchers are exploring ways to design electrode surfaces that encourage bubbles to detach more quickly, preventing them from blocking reactions. Others are experimenting with flowing the liquid electrolyte more aggressively, using motion to sweep bubbles away.
Some of the most intriguing approaches involve changing how the electricity itself is applied. In “pulsed electrolysis,” the current is switched on and off rapidly. During the brief pauses, bubbles have time to dissipate, reducing their buildup and the associated energy losses. “Dynamic operation introduces additional control parameters,” the authors note, opening new possibilities for optimization.
Artificial intelligence is also beginning to play a role, helping scientists analyze complex bubble patterns and optimize operating conditions in ways that would be difficult by hand.
The implications extend far beyond the lab. Global demand for hydrogen is expected to grow dramatically in the coming decades, driven by efforts to cut carbon emissions. Making electrolysis more efficient — even by a small margin — could have an outsized impact on cost and energy use at scale.
“Connecting microscale bubble phenomena to macroscale electrolyzer performance” is therefore critical, the authors argue. In other words, understanding the physics of tiny bubbles could help unlock the full potential of hydrogen as a clean fuel and help improve other gas-evolving electrochemical reactions (or reactors).
Seen this way, the future of green hydrogen may hinge not just on breakthroughs in chemistry or materials science, but on something more fluid and elusive: the behavior of countless small bubbles rising, colliding, and disappearing inside a reactor. Managing them effectively could be the key to turning a promising technology into a practical one.
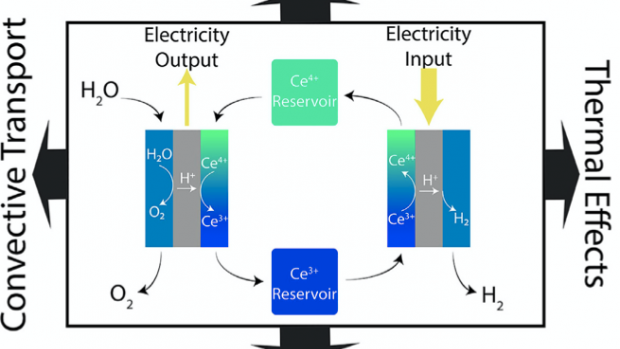
Spatiotemporal Decoupling of Water Electrolysis for Dual-Use Grid Energy Storage and Hydrogen Generation
This research is led by Miguel Modestino, assistant professor of chemical and biomolecular engineering and Yury Dvorkin, assistant professor of electrical and computer engineering.
Increased efforts to curb global warming have led to a drastic surge in the deployment of renewable electricity sources, such as wind and solar power. However, as these sources form a larger fraction of the energy in the grid, their intermittency has started to cause supply instability and large fluctuations in energy prices.
To mediate this challenge, electrochemical energy storage devices such as lithium ion batteries have started to enter the utility scale market. Also gaining traction are redox flow batteries (RFB) because of their long cycle life and power and energy capacity. Despite their promising performance RFB costs remain high due to low energy density of redox energy carriers, which results in large operational costs.
An alternative energy storage method is the production of energy-dense electrofuels, such as hydrogen (H2). H2 has gained significant attention as a promising energy vector for a renewable-rich energy future, given its high gravimetric energy density that makes it desirable for both stationary and mobile applications.
To make electrochemical hydrogen production more competitive, renewable energy sources need to be used with new strategies for electrochemical hydrogen production. The researchers, including Ph.D. students Daniel Frey (lead author) and Jip Kim, show a cerium-mediated decoupled electrolysis system that produces hydrogen and stores energy in the redox couples. They present electrochemical studies to observe the effects of diffusive transport, convective transport, and thermal effects. Following this, the team performed a techno-economic analysis, focusing on the optimization of the system operation and the identification of target operation parameters to achieve hydrogen production at a competitive price.