David Truong
,
Ph.D.
-
Assistant Professor of Biomedical Engineering

Professor Truong works in the area of synthetic and systems biology, where he and his lab focus on building engineered cell therapies by using the techniques of "synthetic genomics". They perform large scale human genome engineering in pluripotent stem cells (iPSCs), which can become any desired cell-type in the body. The lab turns these engineered cells into "off-the-shelf" somatic cells with therapeutic potential, focusing initially on immune cells and neurons. The potential applications of these engineered cells include cancer cell immunotherapy, cell replacement therapy, tissue regeneration, and reducing autoimmune dysfunction.
- B.S. - University of California San Diego
- Ph.D. - The University of Texas at Austin
- Postdoctoral Fellowship - NYU Langone Health
- NIH (NIAID) Director's New Innovator Award (DP2)
- NIAID Small Business Innovative Research Award (SBIR)
- Delil Nasser Award for Professional Development, Genetics Society of America
- NIH (NIGMS) Ruth L. Kirschstein National Research Service Award
- National Science Foundation Graduate Research Fellowship (honorable mention)
Associated Faculty - NYU Grossman School of Medicine - Department of Pathology
Member, Translational Immunology Center
Member, Perlmutter Cancer Center
Founding Team - Neochromosome Inc, a synthetic genomics company
BIGY-7683 (Fall) Biology and Biotechnology for Bioinformatics - Graduate level introduction to the theoretical and applied foundational technologies and techniques used in modern genomics and bioinformatics, e.g., high-throughput sequencing, RNA-seq, GWAS, tumor profiling, etc.
BEGY-9453 (Spring) Engineering Tissue Regeneration - Graduate level overview of current findings, approaches, and theory underlying regenerative medicine and regenerative tissue engineering. The course takes a comprehensive view of the body from molecules, DNA, epigenomics, cell-to-cell communication, tissue physiology, genome engineering, immunology, and systems/synthetic biology.
Research News
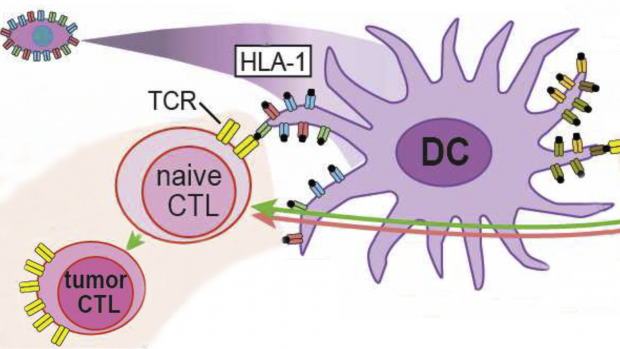
Programmable off-the-shelf dendritic cells as an immunotherapy discovery platform
A new therapeutic era has been ushered in with Adoptive Cell Immunotherapy, which uses patient-harvested T cells genetically engineered against tumor-specific targets. However, the number of addressable cancers is limited by a lack of tumor-specific targets and T cell receptors.
A team led by David M. Truong, Ph.D., assistant professor of biomedical engineering, has been awarded a $1.8 million grant from the National Institute of Allergy and Infectious Diseases (NIAID) under the NIH Director’s New Innovator program, to address this issue by expanding the number of human leukocyte antigen (HLA) restricted T cell receptors (TCRs) and tumor-specific antigens (TSAs), thus transforming the entire immunotherapy pipeline.
To accomplish this, Truong proposes the generation of programmable Dendritic cells (DCs), which are professional antigen presenting cells that function to mature and activate naive T cells. These programmable DCs would facilitate the discovery of new TCRs, the validation of TSAs, and could even be used as “living” vaccines to marshal a patient’s own immune system against infectious disease and cancer.
Truong specifically hopes to produce off-the-shelf DCs pre-engineered to match any HLA haplotype, even rarer haplotypes, that are pre-encoded with any combination of TSAs. Truong’s group will search for and validate “universal” TSAs and TCRs that can be used broadly in TCR therapy for any patient.
More specifically, the group will focus on peptides expressed from regions of the genome which are normally epigenetically silenced, but which are reactivated in tumor cells, e.g., endogenous retroviruses. To further this, the group will continue to develop technology enabling the “writing” of millions of DNA base-pairs in human-induced pluripotent stem cells (iPSCs). Such technology allows for direct customization of the large HLA locus iPSCs in a single step, the introduction of libraries of potential TSAs, as well as integration of synthetic reporter constructs for safer and scalable directed differentiation of iPSCs to DCs.
This research on allogeneic programmed DCs may catalyze a wide variety of immunotherapy applications and expand access to advanced treatments for a greater number of patients.