Researchers discover how Parkinson’s disease affects time perception
A patient undergoes a simple test to measure vision-based time perception, which can be a marker for Parkinson’s disease.
Photo credit: Scientific Reports volume 9, Article number: 19638 (2019) Creative Commons license information
Nearly a million people in the United States have Parkinson’s disease, and by the time they receive a diagnosis, some 60% of the neurons in key portions of the brain controlling movement may have already been lost.
Scientists know that in many cases non-motor symptoms (including perceptual distortions) predate the well-recognized loss of accurate motor control. A new study conducted by Professor S. Farokh Atashzar, who recently joined NYU Tandon, and his colleagues in Canada offers the first systematic investigation of Parkinson’s disease–related misperceptions of time-based upon visual clues. The team developed new tests for these early diagnostic markers and a new means of tracking disease progress free from the confounding artifacts of movement.
They reported promising outcomes from their simple visual test, which measured patients’ time perception using data-driven computational models. Their research, published recently in the Nature journal Scientific Reports, demonstrated for the first time that impaired vision-based time perception can indeed be used as a marker for the disease — bringing hope that the test will aid both early detection and effective long-term monitoring because sensory perception deficits affect 90% of Parkinson’s disease patients.
The four-year study filled in some key knowledge gaps about how Parkinson’s disease degrades the functions of basal ganglia, one of the brain’s control centers for sensorimotor skills. The team demonstrated that Parkinson’s disease significantly diminishes the time perception ability of patients — whether newly diagnosed with few symptoms or far into the disease progression, and whether or not they were using medication or brain stimulation therapy.
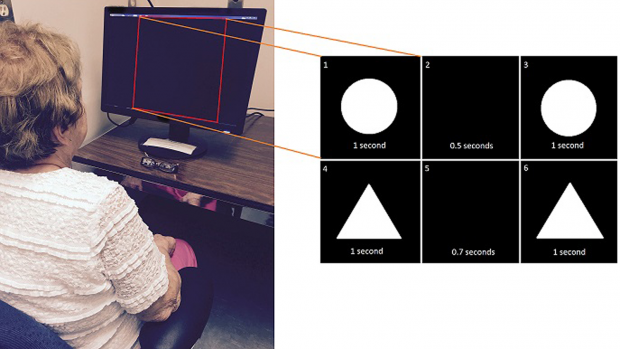
The test was quite simple and could be used in any setting. Participants faced a computer monitor in which a circle and triangle appeared at variable programmed intervals. They estimated the time between the appearance of each blinking object over several iterations, which allowed the researchers to generate robust statistical computational models of perception. When the images flashed at short time intervals, the researchers detected no meaningful differences between the patients and the control group. But when they spaced the images at intervals of one second or more — the time frame controlled by the basal ganglia — consistent abnormalities became apparent.
The study also suggested impaired visual perception of time even for participants using brain stimulation therapy or Levodopa, a medicine used to treat Parkinson’s disease. The team also found an interesting correlation between impaired perceptual perception and motor impairment caused by disease progression.
The researchers plan to continue developing the tool and computational models for clinical and research purposes — perhaps adding haptics, auditory, or other sensory perception tests.
The lead author of “Differential Temporal Perception Abilities in Parkinson’s Disease Patients Based on Timing Magnitude” is Matthew Bernardinis, who was a master’s degree student co-advised by Atashzar, Rajni V. Patel, and Mandar S. Jog, both of the University of Western Ontario.
Atashzar joined NYU Tandon in 2019 as an assistant professor appointed to the Department of Electrical and Computer Engineering as well as the Department of Mechanical and Aerospace Engineering. Previously, he was a senior postdoctoral scientist in the Department of Bioengineering at the Imperial College London in the United Kingdom, sponsored by the Natural Sciences and Engineering Research Council (NSERC) of Canada. From February 2017 to August 2018, he served as a postdoctoral research associate at Canadian Surgical Technologies and Advanced Robotics (CSTAR) center. His research employs artificial intelligence, advanced control systems, signal processing algorithms, smart wearable mechatronic modules, and human-robot interaction systems to augment human capabilities and understand the functions of the human nervous system.



