Polymer Membranes for Large-Scale Energy Conversion and Storage
Speaker:
Michael A. Hickner
Department of Material Science and Engineering, Pennsylvania State University
Abstract:

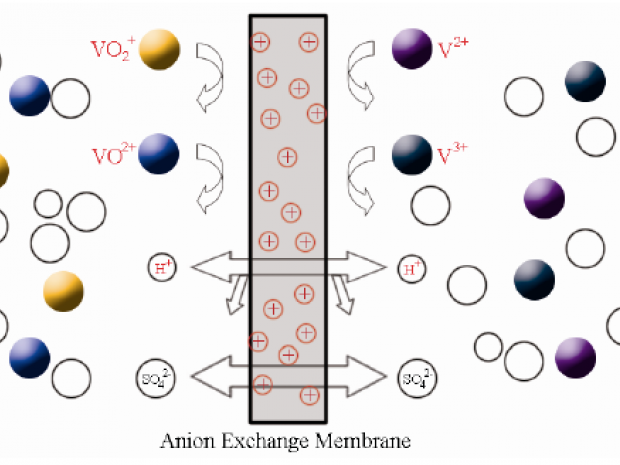
New polymer membranes are needed to advance energy storage and conversion technologies for distributed and grid-scale applications. We have recently demonstrated new ion-conducting polymer membranes that have achieved excellent performance and long-lifetime stability in vanadium redox flow batteries, a leading technology candidate for deployment in renewable power networks and grid-scale energy storage systems with sizes ranging from 10s to 100s of megawatts. By tuning the nanoscopic self-assembly of the ionic domains in the polymers, we are able to increase the cycle life of the device by impeding vanadium ion transport through the membrane while facilitating high conductivity in the electrolyte to maintain the battery current density. For instance, by decreasing the vanadium permeability of the membrane by a factor of two, we have been able to double the lifetime of the device, which provides significant life-cycle cost savings. We have also demonstrated membranes with nearly zero vanadium permeability that show 100 % coulombic efficiency in flow battery charge-discharge cycling tests. Currently, we are working on demonstrating these membranes over 100s of charge-discharge cycles.
In addition to optimizing the chemical structures of polymeric membranes, physical manipulations, such as surface patterning, can be employed to improve the performance in devices. We have used photopolymerization and a stereolithographic patterning 3D printing process to develop surface-patterned anion exchange membranes that have low resistance compares to their flat, unpatterned counterparts. The lower resistance of the patterned membranes is due to the low parallel resistance of the patterned areas and is quantified through a mathematical model that can be used as a predictive tool for optimizing the membranes.
This talk will show how polymers with new chemical structures and physical patterning can be applied to different types of batteries and other electrochemical devices. Common design principles and considerations for fabricating new ion exchange membranes for energy processes will be discussed.
- 10:30 Refreshments
- 10:45–12:00 Talk

